Look at the table of electron configurations in Appendix c. Which elements have configurations that are exceptions
Question:
Look at the table of electron configurations in Appendix c. Which elements have configurations that are exceptions to the aufbau principle? Propose a reason why these elements have these exceptions.
Data from appendix c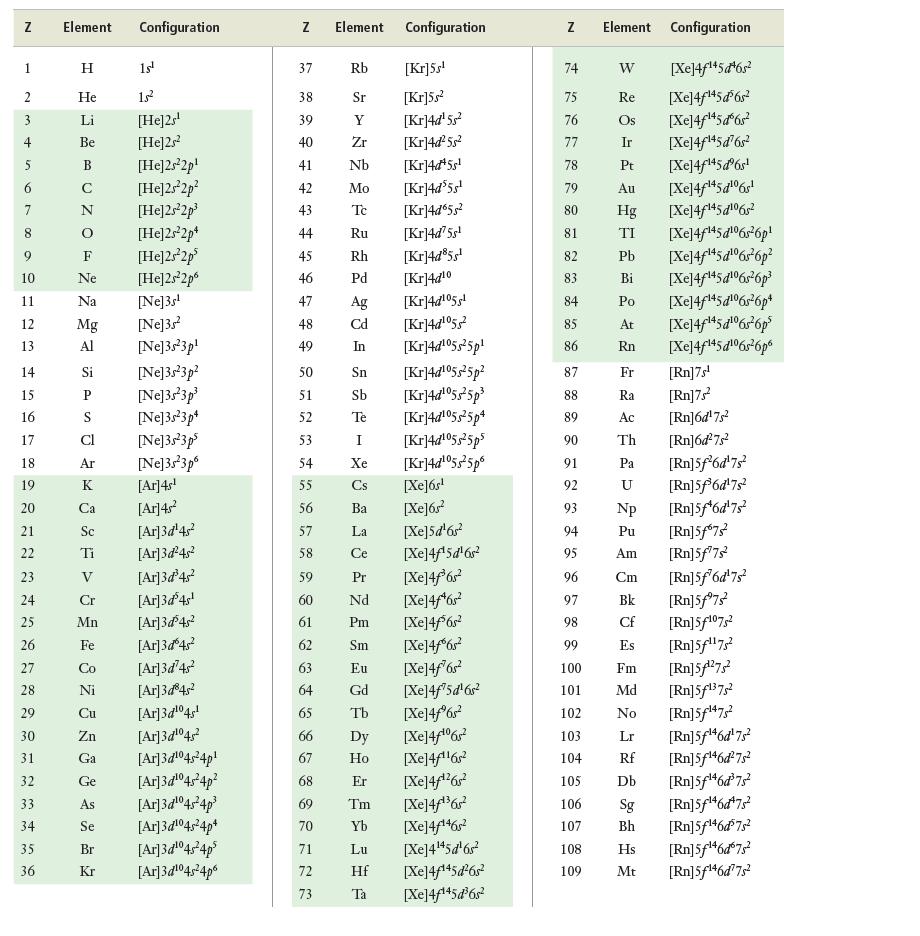
Transcribed Image Text:
N 1 2 + 5 6 7 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 Element Configuration H 15¹ He 15² Li [He]2s¹ Be [He]2s² [He]2s²2p¹ [He]2s²2p² [He]2s²2p³ [He]2s²2p* [He]2s²2p³ [He]2s²2p6 [Ne]3s¹ [Ne]3s² [Ne]3s²3p¹ AUZO Z Z Ne Na Mg Al Si P S Ar K Ca Sc Ti V Cr Mn Fe Co [Ne]3s²3p² [Ne]3s 3p³ [Ne]3s²3p4 [Ne]3s 3p5 [Ne]3s²3p6 [Ar]4s¹ [Ar]4s² [Ar]3d¹4s² [Ar]3d²4s² [Ar]3d³4s² [Ar]3d³4s¹ [Ar]3d³45² [Ar]3d 4s² [Ar]3d¹4² Ni [Ar]3d³45² Cu [Ar]3d¹⁰45¹ Zn [Ar]3d¹⁰45² Ga [Ar]3d¹04s²4p¹ Ge [Ar]3d¹⁰45²4p² As [Ar]3d¹04s²4p³ Se [Ar]3d¹04s²4p+ Br [Ar]3d¹04s²4p³ Kr [Ar]3d¹04s²4p6 N 37 38 39 40 41 42 43 44 45 46 7 48 49 50 47 50 Element Configuration 72 75 Rb [Kr]5s¹ Sr [Kr]55² Y [Kr]4d¹5s² Zr [Kr]4d²5s² Nb [Kr]4d¹5s¹ Mo [Kr]4d³5s¹ Tc [Kr]4d65s² Ru [Kr]4d' 5s¹ Rh [Kr]4d³5s¹ Pd [Kr]4d10 [Kr]4d¹05¹ [Kr]4d¹055² [Kr]4d¹055²5p¹ [Kr]4d¹05s²5p² [Kr]4d¹05s²5p³ [Kr]4d¹05525p4 [Kr]4d¹05s²5ps [Kr]4d¹055²5p6 73 Ag Cd In 51 52 53 54 55 56 Ba 57 La 58 Ce 59 Pr 60 Nd 61 Pm 62 Sm 63 Eu 64 65 Sn Sb Te [Xe]6s¹ [Xe]6s² [Xe]5d¹6s² [Xe]4f¹5d¹6s² [Xe]4far [Xe]4f¹6s² [Xe]4f6r2 [Xe]4f6s? [Xe]4f762 [Xe]4f¹5d¹6s² [Xe]4f®6? 66 Dy [Xe]4/06r 67 Ho [Xe]4f1162 68 Er [Xe]4f*26r? 69 Tm [Xe]4f36r2 70 Yb [Xe]4f1462 71 Lu [Xe]4145d6r Hf [Xe]4f¹45d²6s² Ta [Xe]4f¹45d³6s² I Xe Cs Gd Tb Z 74 75 76 77 78 79 80 81 82 83 84 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 100 101 102 103 104 105 106 107 108 109 Element W Re Os Ir Pt Au Hg TI Pb Bi Po At Rn Rf Db Configuration [Xe]4f145 đ6g? [Xe]4f14562 [Xe]4/145d6s? [Xe]4f145d6s? [Xe]4f¹45dº6s¹ [Xe]4f**5¢06r! [Xe]4f145g16g? Sg Bh [Xe]4f45d!06r6p! [Xe]4f**5d!06r6p2 [Xe]4f145¢106s6p* [Xe]4f145¢106s26p* [Xe]4f**5¢!6r6p* [Xe]4f**5d106r6p Fr Ra Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md [Rn]5f¹³75² No [Rn]5f¹47² Lr [Rn]5f¹46d¹75² [Rn]5f¹46d²75² [Rn]5f¹46d³75² [Rn]5f¹46d75² [Rn]5f¹46d³75² Hs [Rn]5f¹46d675² Mt [Rn]5f¹46d775² [Rn]7s¹ [Rn]7s² [Rn]6d¹7s² [Rn]6d²7s² [Rn]5f6d¹7s [Rn]5f³6d¹7s² [Rn]5f¹6d¹7s² [Rn]5f673² [Rn]5f¹7s² [Rn]5f6d¹7s² [Rn]5f7s² [Rn]5f¹07,² [Rn]5f¹¹75² [Rn] 5¹27²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The elements that have configurations that are exceptions to the ...View the full answer

Answered By

Ariz Azad
I love explaining things to people. Sharing my knowledge with others is one of the rare things that gives me immense pleasure. I have been a tutor on Chegg for more than a year now for Finance. Nothing beats the satisfaction of getting a thumbs up from students for your answer. Maintaining quality in teaching is one of the things I have learned through experience.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
The CICA Handbook is available to most post-secondary students through their institutions subscription. Visit edu.knotia.ca to complete this exercise. (You may need to complete this exercise on...
-
Look again at the table of post-graduation plans for the senior class in Exercise 27. a) Find the conditional distributions (percentages) of plans for the white students. b) Find the conditional...
-
Write a C++ program with recursion to print the following pattern for any odd number greater than or equal to 5. Note: the last row must be filled with digits from your ID in case the pattern is...
-
Presto Corp. has collected the following data concerning its maintenance costs for the past 6 months. Compute the variable and fixed cost elements using the high-lowmethod. July August September...
-
RMC, Inc., is a small firm that produces a variety of chemical products. In a particular production process, three raw materials are blended (mixed together) to produce two products: a fuel additive...
-
When parties must arbitrate their disputes?
-
Refer to the data given in Problem 4.34 and suppose that the expected activity costs are reported as follows (all other data remain the same): Other overhead activities: Activity Cost Purchasing...
-
2. Prove the following theorem: A sequence of functions (fr) defined on ACR converges uniformly on A if, and only if, Ve > 0 there exists an N = N such that |fn(x) fm(x)| < for all m, n N and all x =...
-
Distinguish between the terms core electrons and valence electrons.
-
From the list of atoms and ions given, identify any pairs that have the same electron configurations and write that configuration: Na + , S 2-, Ne, Ca 2+ , Fe 2+ , Kr, I - .
-
A point source with volume flow Q = 30 m3/s is immersed in a uniform stream of speed 4 m/s. A Rankine half-body of revolution results Compute (a) The distance from the source to the stagnation point;...
-
[ The following information applies to the questions displayed below. ] The following information pertains to the inventory of Parvin Company for Year 3 : January 1 Beginning inventory 3 0 0 units @...
-
Let L = {a'bij). How many strings of length less than or equal to 3 are in L? Answer:
-
Describe 360-degree evaluations and explain their importance. Define Forced Distribution and provide two advantages and two disadvantages List and explain the steps of the Performance Management...
-
Multiply. (-24)(-2)
-
During the first month of operations ended July 31, YoSan Inc. manufactured 2,400 flat panel televisions, of which 2,000 were sold. Operating data for the month are summarized as follows: Sales...
-
Using the Internet, research two organizations that offer certifications in forensic accounting. Visit each organizations web site and explore the nature of the certification and how it is obtained...
-
Copy and complete the statement. 3800 m ? km =
-
Identify which of the following compounds is expected to have the larger heat of combustion:
-
Draw each of the following compounds: (a) 2,2,4-Trimethylpentane (b) 1,2,3,4-Tetramethylcycloheptane (c) 2,2,4,4-Tetraethylbicyclo [1.1.0] butane
-
Sketch an energy diagram that shows a conformational analysis of 2,2-dimethylpropane. Does the shape of this energy diagram more closely resemble the shape of the energy diagram for ethane or for...
-
On December 31, 2024, Crane Landscapings common shares were trading for $15, and the company had the following shareholders equity accounts: Common Shares (no par value, unlimited authorized, 970,000...
-
The operating revenues and expenses of Panawin Enterprises for 2 0 2 1 are below:There are four additional transactions that impact the income statement: Machinery with a balance sheet ( book ) value...
-
Laker Company reported the following January purchases and sales data for its only product. The Company uses a perpetual inventory system. For specific identification, ending inventory consists of...

Study smarter with the SolutionInn App


