Nitrogen is capable of forming single, double, or triple bonds, and the figure that follows shows the
Question:
Nitrogen is capable of forming single, double, or triple bonds, and the figure that follows shows the potential energy as a function of internuclear distance for each of these types of bonds. Match the three curves in the figure (A, B, and C) to the three types of bonds. Explain your reasoning.
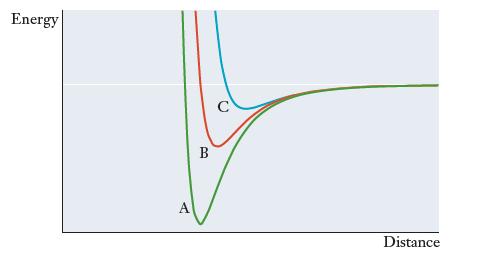
Transcribed Image Text:
Energy B C Distance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Triple bonds are stronger and shorter than double bonds w...View the full answer

Answered By

Parvesh Kumar
I am an experienced Mathematics and Statistics tutor with 10 years of experience teaching students and working professionals. I love teaching students who are passionate to learn subjects or wants to understand any mathematics and statistics concept at graduation or master’s level. I have worked with thousands of students in my teaching career. I have helped students deal with difficult topics and subjects like Calculus, Algebra, Discrete Mathematics, Complex analysis, Graph theory, Hypothesis testing, Probability, Statistical Inference and more. After learning from me, students have found Mathematics and Statistics not dull but a fun subject. I can handle almost all curriculum of mathematics. I did B.Sc (mathematics), M.Sc (mathematics), M.Tech (IT) and am also Gate (CS) qualified. I have worked in various college and school and also provided online tutoring to American and Canadian students. I look forward to discussing with you and make learning a meaningful and purposeful
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
The figure that follows shows ball-and-stick drawings of three possible shapes of an AF3 molecule. (a) For each shape, give the electron-domain geometry on which the molecular geometry is based. (b)...
-
A 230.0-g object on a spring oscillates left to right on a frictionless surface with a frequency of 2.00 Hz. Its position as a function of time is given by x = (8.00 cm) sin ( t.? (a) Sketch a graph...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
1Describe in a simplified manner the flow of information from gene to protein
-
Are there problems with scenario analysis? Define simulation analysis, and discuss its principal advantages and disadvantages. Shrieves Casting Company is considering adding a new line to its product...
-
The LawnPlus Fertilizer Company distributes fertilizer to various lawn and garden shops. The company must base its quarterly production schedule on a forecast of how many tons of fertilizer will be...
-
The company is committed to ethical conduct and has no tolerance for fraud and unethical behavior. There are some concerns about abuses in your department. Do you know anything about the concerns I...
-
The teenage daughter of a recently deceased movie star inherited a number of items from her famous fathers estate. Rather than convert these assets to cash immediately, her financial advisor has...
-
How would a data analyst evaluate a conference call to get a feel for the sentiment of management?
-
The N 5 + cation has been synthesized and studied. Consider the possible Lewis structure below. Indicate the hybridization expected for each nitrogen atom and the expected bond angles. Assuming that...
-
Nitrogen triiodide, NI 3 (s), is unstable and will spontaneously detonate to form a bright purple cloud of nitrogen and iodine gases accompanied with a loud bang, which suggests a release of energy....
-
The Robinson-Patman Act prohibits price discrimination. What were the ethical reasons that motivated the passage of this federal statute?
-
Gwen starts a job with a salary of $46,000 and gets a 4% raise every year. How much will she have made in salary in total over the course of her 35 years on the job?
-
Define the following terms 1. Covered interest arbitrage 2. Uncovered interest arbitrage 3. Hedging 4. Forward discount
-
As of today if you are a business manager would you take a fixed interest or floating interest loan? Explain why and how this decision may change?
-
Working at Burger in a Box, you are paid $25 each week plus $6 per hour. This week, you want your pay to be $133. How many hours should you work to earn enough?
-
4. ______________ shapes the culture for process improvement. a. stability b. standardize c. jidoka d involvment
-
Case Development began operations in December 2011. When property is sold on an installment basis, Case recognizes installment income for financial reporting purposes in the year of the sale. For tax...
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
Atomic emission experiments of a mixture show a calcium line at 422.673 nm corresponding to a 1 P 1 1 S 0 transition and a doublet due to potassium 2 P 3/2 2 S 1/2 and 2 P 1/2 2 S 1/2 transitions...
-
Consider the 1s np 3 P 1s nd 3 D transition in He. Draw an energy-level diagram, taking the spin-orbit coupling that splits terms into levels into account. Into how many levels does each term split?...
-
Calculate the transition dipole moment, for a transition from the 1s level to the 2p z level in H. Show that this transition is allowed. The integration is over r, θ, and . Use for the...
-
If 10-year interest rates are at 3%...and its volatility is at 100 bps...how many standard deviations away from a yield of 1% are you? If you increase volatility to 200 bps does it become more or...
-
On January 1, 2XX0, Showy Animals Corp. purchased 100% of the outstanding common stock of Armadillo Shells Inc. On the date of the acquisition, Armadillo Shells' identifiable net assets had fair...
-
How does symbolic interactionism, originating from the works of George Herbert Mead and later developed by scholars like Herbert Blumer, analyze society through the lens of subjective meanings and...

Study smarter with the SolutionInn App


