Starting with 238 U and ending with an isotope of Pb, write a decay scheme in which
Question:
Starting with 238U and ending with an isotope of Pb, write a decay scheme in which every nuclide emits only an particle. Compare your scheme to the decay series in Figure 14.5 and suggest a reason why β– emissions must occur in the 238U decay series.
Figure 14.5
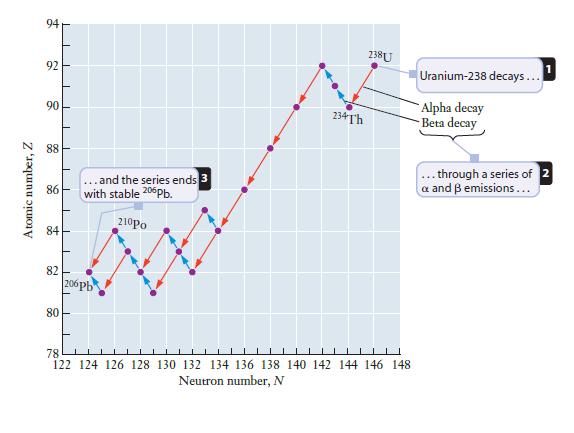
Transcribed Image Text:
Atomic number, Z 94 92 90 88 86 84 82 80 E 1 I I I II ... and the series ends 3 with stable 206Pb. 210Po 206pb 4444 234Th 238 U ||▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬ 78 LLL 122 124 126 128 130 132 134 136 138 140 142 144 146 148 Neutron number, N Uranium-238 decays... Alpha decay Beta decay ... through a series of 2 a and B emissions...
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The decay scheme of 238U to Pb with only alpha decay is as follows 238U 234Th 230Th 226Ra 222Rn 218P...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
A block with mass m is attached to an ideal spring that has force constant k. (a) The block moves from x, to x" where x, > x,. How much work does the spring force do during this disparagement? (b)...
-
In a special double issue of Time magazine, the cover story featured Pope John Paul II as Man of the Year (26 December 19942 January 1995, pp. 7476). As part of the story, Time reported on the...
-
The two most common isotopes of uranium are 235U and 238U. (a) Compare the number of protons, the number of electrons, and the number of neutrons in atoms of these two isotopes. (b) Using the...
-
What are the possible sources of error in this project?
-
Research the latest developments in the class action lawsuit involving women managers versus Wal-Mart stores and report your findings to the class.
-
Chris Fisher, owner of an Ohio firm that manufactures display cabinets, develops an 8-month aggregate plan. Demand and capacity (in units) are forecast as follows: The cost of producing each unit is...
-
The Korvette concept was started and run by one person and his group of friends. How could its failure have been avoided? Was the problem one of strategy (overexpansion), or was it organizational?...
-
Trader Cycles manufactures chainless bicycles. On March 31, Trader Cycles had 220 bikes in inventory. The company has a policy that the ending inventory in any month must be 30% of the following...
-
Warranties normally: Multiple select question. have indefinite lives. guarantee repair or replacement. represent liabilities. cover a specific time period.
-
Write a nuclear equation for the type of decay each of these unstable isotopes is most likely to undergo. (a) 19 Ne (b) 230 Th (c) 82 Br (d) 212 Po
-
The following successive decays occur, starting with 24 F, which has a half-life of 0.3 s, and ending at a stable isotope: (a) Identify each of the decay products. (b) Using the chart of the...
-
Nana found some coins under her sofa pillows. There were equal numbers of nickels and quarters and twice as many half-dollars as quarters. If she found $2.60 in all, how many of each denomination of...
-
The US Department of Homeland Security (DHS) Doctrine defines internal risks as follows Financial stewardship Malicious activity in cyberspace Personnel reliability Systems reliability
-
2. A wind farm off the cost of Scotland consists of 60 wind turbines, each expected to generate 3 MW of electrical energy when the wind speed is between 15-25 m/s (at gale strength), with a standard...
-
Describe each of the research methods questionary, interviews, observation, and documents and images and when it might be best to employ it, from Martyn Denscombe's book.
-
How do facilities ensure their cost management practices align with regulatory requirements?
-
What mechanisms can organizations implement to ensure ethical behavior and social responsibility among their employees? Explain
-
1. We have 800 m3 of natural gas at 235 kPa at 30C. What is the temperature if the volume is changed to 1200m3 at 215 kPa? 2. We have 1400 L of nitrogen at 135 kPa at 54C. What is the temperature if...
-
The Strahler Stream Order System ranks streams based on the number of tributaries that have merged. It is a top-down system where rivers of the first order are the headwaters (aka outermost...
-
Solve Prob. 938 for point B, which is located on the outer surface of the pipe. 12 in. 60 lb 30 12 in. B.
-
The wide-flange beam is subjected to the 50-kN force. Determine the principal stresses in the beam at point A located on the web at the bottom of the upper flange. Although it is not very accurate,...
-
Solve Prob. 940 for point B located on the web at the top of the bottom flange. 50 kN B. -1 m- 3 m 12 mm 10 mm- 250 mm 12 mm 200 mm
-
Suppose you (each one of you in your group) own a power generation plant. The plants are not very similar. Some of you own plants that release more carbon dioxide than others. And some of you own...
-
You have the following data: Earnings before interest and taxes Depreciation Taxes operating cash flow in $ is, Your Answer: Answer $700 $150 $54 Round your answer to 2 decimal points; example 1.12
-
A Treasury bill has a face value of 100K, 177 days to maturity, and is selling for $98 thousand. What is the effective annual yield on this bill? Enter answer in percents, accurate to two decimal...

Study smarter with the SolutionInn App


