An aqueous solution of hydrazine, N 2 H 4 , can be prepared by the reaction of
Question:
An aqueous solution of hydrazine, N2H4, can be prepared by the reaction of ammonia and sodium hypochlorite.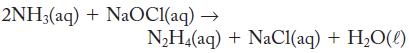
What is the theoretical yield of hydrazine, in grams, prepared from the reaction of 50.0 mL of 1.22 M NH3(aq) with 100.0 mL of 0.440 M NaOCl(aq)?
Transcribed Image Text:
2NH3(aq) + NaOCl(aq) → N₂H4(aq) + NaCl(aq) + H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To calculate the theoretical yield of hydrazine we need to use the stoichiometry of the balanced equ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
What mass of silver chloride can be prepared by the reaction of 100.0 mL of 0.20 M silver nitrate with 100.0 mL of 0.15 M calcium chloride? Calculate the concentrations of each ion remaining in...
-
Ethylammonium nitrate, CH 3 CH 2 NH 3 NO 3 , was the first ionic liquid to be discovered. Its melting point of 12 C was reported in 1914, and it has since been used as a nonpolluting solvent for...
-
You are looking to help a small municipality finance their drinking water treatment facility with the installation of UV (ultraviolet) disinfection. The total cost of the project is estimated to be...
-
Consider the SDE for the spot rate r t Suppose the parameters a, ?, ? are known, and that as usual, W t , is a Wiener process. (a) Show that (b) What do these two equations imply for the conditional...
-
In Kazakhstan, the demand curve 12 of dairy products is q = kp0.6 for some positive constant k. What is the elasticity of dairy products in Kazakhstan?
-
Calculate the inductance of a 2000 -winding cylindrical solenoid that is \(0.20 \mathrm{~m}\) long if the radius of each winding is \(0.030 \mathrm{~m}\).
-
Fungus Audio Works Inc. warrants its products for one year. The estimated product warranty is 3% of sales. Assume that sales were $680,000 for January. In February, a customer received warranty...
-
QUESTION 3 (20 MKS) a) Obtain the time-independent Schrodinger Wave equation from the time dependent equation. [10 mks] b) Solve the time-independent Schrodinger equation given that,(x) = Aekx [10...
-
How do you calculate the percentage of change in sales from 20X8 to 20X9? This will go in cell D7? Show the formula. Kretsmart Common-Size Income Statements For the Years Ended December 31, 20'X'9,...
-
Tin(II) fluoride (stannous fluoride) is added to toothpaste as a convenient source of fluoride ion, which is known to help minimize tooth decay. The concentration of stannous fluoride in a particular...
-
A 2.64-g sample of Ba(OH) 2 is dissolved in water to form 250.0 mL solution. Th is solution is titrated with 0.0554 M H 2 SO 4 . It takes 33.4 mL of the acid solution to neutralize 30.0 mL of the...
-
Verify that if \(a=p_{a}+i q_{a}\) and \(b=p_{b}+i q_{b}\) are two complex numbers, then the product of the two is the complex number. Get real and imaginary part.
-
In February 2018, Judy Judd gave her son Steven a Mercedes for his graduation gift. The automobile had a basis of \(\$ 55,000\) to Judy but was worth \(\$ 85,000\) at the time of the gift. Judy paid...
-
Adam Armstrong, age 60 and single, earned \(\$ 71,000\) during 2018 . He contributed \(\$ 9,000\) to the United church, paid \(\$ 16,000\) in federal income taxes, \(\$ 3,400\) in state income tax,...
-
In 2020, flow A was $1,000 billion, flow C was $250 billion, flow B was $650 billion, and flow E was $50 billion. Calculate investment. FACTOR MARKETS A ( HOUSEHOLDS FIRMS D ( GOVERNMENTS B GOODS...
-
You are residence director at Expensive Private University. Enrollment at your school has been declining because of repeated publicity about excessive drinking among the students. Last year, 23 were...
-
Hatcher Village, which operates on the calendar year, issued a 5-year, 8%, $100,000 note to the Bank of Hatcher on January 5, 20X4. The proceeds of the note were recorded in a Capital Projects Fund....
-
An analogy used in relation to venture opportunity screening makes reference to "caterpillars" and "butterflies." Briefly describe the use of this analogy.
-
Bobbie Singh provides writing services for small businesses. He blogs for companies that need professionally written content. His business records at November 15, 2023, are shown below: During the...
-
In the absence of turbulent mixing, the partial pressure of each constituent of air would fall off with height above sea level in the Earths atmosphere as P i = P i 0 e -M,g/RT where P i is the...
-
Propose a mechanism for the following transformation: . 1) Excess LA, 2) H20
-
Propose a mechanism for the following transformation: . 1) Excess MeMgBr 2) H20
-
Account information Accrued Liabilities $ 23,000 Accounts Payable $ 36,000 Long-term Note Payable Accounts Receivable, net Cash 100,500 Retained Earnings ? 100,000 Interest Expense 10,850 35,000...
-
The hospital has several specialized departments like Cardiology. Gynecologic, Orthopedics, Pediatrics, ENT etc. OPD is another independent department. A doctor is only associated with one...
-
As you organize a dinner assembly for your closest friends at a restaurant in your city, you find yourself searching for the perfect place to dine. After much contemplation, you decide to take your...

Study smarter with the SolutionInn App


