Find the following elements in the periodic table, identify which block they are in, and give their
Question:
Find the following elements in the periodic table, identify which block they are in, and give their electron configurations by using the structure of the periodic table rather than consulting Table 7.3.
(a) Br
(b) Ti
Table 7.3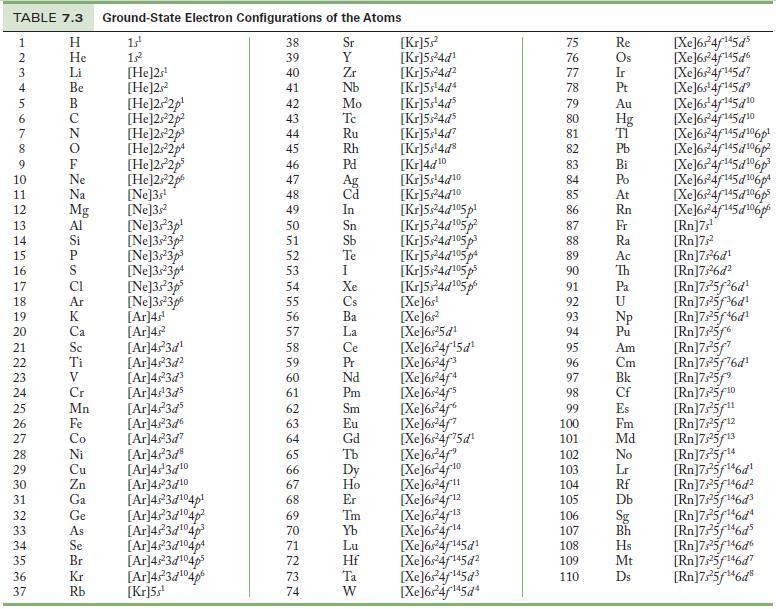
Transcribed Image Text:
TABLE 7.3 Ground-State Electron Configurations of the Atoms 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 HIJABUZOLASJE E>282556822 Η He Li Be Ne Na Mg Al Si P Ar Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb 1s¹ 15² [He]2s¹ [He]2s2² [He]2s²2p¹ [He]2s²2p² [He]2s²2p³ [He]2s22p4 [He]2s²2p³ [He]2s22p [Ne]3s¹ [Ne]3s² [Ne]3s 3p [Ne]3s23p² [Ne]3s 3p³ [Ne]3s 3p4 [Ne]3s 3p³ [Ne]3s 3p6 [Ar]4s¹ [Ar]4² [Ar]4s²3d¹ [Ar]4s²3d² [Ar]4s²3d³ [Ar]4s¹3d³ [Ar]4s²3d5 [Ar]4s²3d6 [Ar]4s²3d7 [Ar]4s²3d8 [Ar]4s¹3d10 [Ar]4s²3d10 [Ar]4s23d¹04p¹ [Ar]45³3d¹04p² [Ar]45²3d¹04p³ [Ar]4s²3d¹04p4 [Ar]4s23d¹04p³ [Ar]4523d104p [Kr]5s¹ 38 39 45454647 48 49 50 51 52 53 4 55 56 >N22222232823 fáa fA> 62 64 65 66 68 69 70 71 72 73 74 40 41 42 43 54 57 58 59 60 61 63 Sr 67 Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Hf Ta W [kr]5s² [kr]5s²4d¹ [Kr]5s²4d² [kr]5s¹4d4 [kr]5s¹4d³ [kr]5s²4d³ [kr]5s¹4d² [kr]5s¹4d8 [kr]4d10 [kr]5s¹4d10 [kr]5s24d10 [kr]5s24d¹05p¹ [kr]524d¹05p² [kr]5s²4d¹05p3 [kr]5s²4d¹05p4 [kr]5s24d¹05ps [Kr]524d¹05p [Xe]6s¹ [Xe]6s² [Xe]6s¹5d¹ [Xe]6s¹4f¹5d¹ [Xe]6s¹4f³ [Xe]6s4f4 [Xe]6s²4f's [Xe]6347% [Xe]6s¹4f [Xe]6s¹4f75d¹ [Xe]6s¹4f" [Xe]64f10 [Xe]64f11 [Xe]6s¹4f12 [Xe]634f13 [Xe]6s²4f14 [Xe]6s¹4f¹45d¹ [Xe]6s²4f145d² [Xe]654f145d3 [Xe]6s²4f¹45d4 75 76 GLASSLISTEN 77 78 79 81 82 83 85 86 80 Hg 87 88 89 90 91 92 94 95 96 97 98 99 100 101 102 103 104 105 106 107 93 Np 108 _ROHAUTBALHLHA&DAU SHJGG BLTW LL H SAM B 109 Re 110 Os Ir Pt Au Bi Po At Rn Fr Ra Ac Th Pa Pu Am Cm Bk Cf Es Fm Md No Lr Rf ᎠᏏ Bh Hs Mt Ds [Xe]64f145d5 [Xe]64/145d6 [Xe]63*4f45d7 [Xe]6s¹4f¹45d⁹ [Xe]6s¹4f¹45d10 [Xe]6r4f145d10 [Xe]6s¹4f145d¹06p [Xe]6s 4f¹45d¹06p [Xe]634f45d6p [Xe]6s²4f¹45d¹06p4 [Xe]64f5d106p [Xe]6®4f145d6p [Rn]7s¹ [Rn]7s² [Rn]7s²6d¹ [Rn]7s²6d² [Rn]7s²5f²6d¹ [Rn]7,²5f³6d¹ [Rn]7,²5f¹6d¹ [Rn]7,25f6 [Rn]7s¹5f7 [Rn]7,25f76d¹ [Rn]7s²5f⁹ [Rn]7,25f10 [Rn]7,25/¹¹ [Rn]7,25f¹2 [Rn]7,25f¹3 [Rn]7,25/14 [Rn]7s²5f¹46d¹ [Rn]7s²5f¹46d² [Rn]7s²5f¹46d³ [Rn]7,25f¹6d [Rn]7s²5f¹46d5 [Rn]7,25f¹46d6 [Rn]7s25f¹46d7 [Rn]7,25f¹46d
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a b ...View the full answer

Answered By

Sandhya Sharma
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
119+ Reviews
214+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Find the following elements in the periodic table, identify which block they are in, and give their electron configurations by using the position in the periodic table rather than consulting Table...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Using periodic trends, arrange the following elements in order of increasing atomic radius: O, P, S.
-
} S 1995 the # of Farms Century, data year 1935 1990 19.50 Aumcon people living on declined steadily during the shown by the Follow g as (in milion of persons) from 1935 19.55 1960 1965 11975 1980...
-
Classification of casts, service sector Consumer Focus is a marketing research firm that organizes focus groups for consumer-product companies. Each focus group has eight individuals who are paid $50...
-
The Home Depot, Inc., financial statements appear in Appendix A at the end of this textbook. Use these statements to answer the following questions: a. What is the total dollar value of the companys...
-
Show that the variance of the $i^{\text {th }}$ run in a $2^{3}$ design with two replications is \[\frac{\left(y_{i 1}-y_{i 2} ight)^{2}}{2}\] where $y_{i j}$ is $j^{\text {th }}$ observation in...
-
The Hernandez Manufacturing Company has two producing departments, machining and assembly. Mr. Hernandez recently automated the machining department. The installation of a CAM system, together with...
-
Suppose that the S&P 500, with a beta of 1.0, has an expected return of 13% and T-bills provide a risk-free return of 4%. 13 a. What would be the expected return and beta of portfolios constructed...
-
Use the aufbau procedure to obtain the electron configuration and orbital diagram for atoms of the following elements. (a) Be (b) B (c) Ne (d) Rb
-
List the elements O, S, and Al in order of increasing size.
-
You are the librarian for a large firm with a large library. On a piece of graph paper, illustrate how you would organize the library. Include placement of the following: a. Statutes b. Reporters c....
-
Oa. Equity -are cash and other assets expected to be converted to cash, sold, or consumed either in a year. O b. Non current liabilities Oc Current assets Od. Current liabilities Oe. Non current...
-
Consider a particle in a 1D half-infinite well. The potential in region III is infinite and is zero in region I. In this case however, the potential is not infinite in region II but has a constant...
-
Perpetual Inventory Using LIFO Beginning inventory, purchases, and sales for Item GY9 are as follows: Mar. 1 Inventory 365 units at $24 8 Sale 305 units 15 27 Purchase 510 units at $26 Sale 325 units...
-
Al Avocadoand Juan Tammeraare the two main bartenders at Pepe's Saloon in Key West, Florida. Anticipating crowds of students during Spring Break they order twice their usual amount of Crazy Lady...
-
The purpose of this lab exercise is to familiarize you with simple arithmetic, relational, and if/else operations and the creation of a simple GUI interface involving Button and TextField components....
-
If an industry created both external benefits and external cost, would a tax or subsidy be appropriate? Why would someone be hurt by either one?
-
What are three disadvantages of using the direct write-off method?
-
Draw and name the structure of the aldohexose that is epimeric with d-glucose at each of the following positions: (a) C2 (b) C3 (c) C4
-
The same product is obtained when either d-altrose or d-talose is treated with sodium borohydride in the presence of water. Explain this observation.
-
The same product is obtained when either d-allose or l-allose is treated with sodium borohydride in the presence of water. Explain this observation.
-
Brooks Plumbing Products Incorporated ( BPP ) manufactures plumbing fixtures and other home improvement products that are sold in Home Depot and Walmart as well as hardware stores. BPP has a solid...
-
On January 1 , 2 0 X 3 , Poke Corporation acquired 2 5 percent of the outstanding shares of Shove Corporation for $ 1 0 0 , 0 0 0 cash. Shove Company reported net income of $ 7 5 , 0 0 0 and paid...
-
Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company has two manufacturing departments - Molding and Fabrication. It started, completed, and...

Study smarter with the SolutionInn App


