From the data presented in Figure 12.11, determine which has the more positive enthalpy of solution: NaCl
Question:
From the data presented in Figure 12.11, determine which has the more positive enthalpy of solution: NaCl or NH4Cl. Explain.
Figure 12.11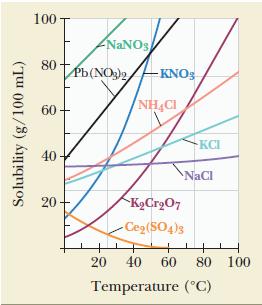
Transcribed Image Text:
Solubility (g/100 mL) 100 80 60 40 20 NaNO3 Pb(NO3)2KNO3 NHC KCrO7 -Ce(SO4)3 KCI 60 NaCl 20 40 Temperature (C) 80 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
NH 4 Cl has the more po...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
From the data presented in Figure 12.11, determine which has the more positive enthalpy of solution: NaCl or KNO 3 . Explain. Figure 12.11 Solubility (g/100 mL) 100 80 60 40 20 NaNOs Pb (NO3)2KNO3...
-
Reconsider the Special Products Company problem presented in Section 1.2. Although the company is well qualified to do most of the work in producing the iWatch, it currently lacks much expertise in...
-
The data presented in Table suggest that the corporate form suffered a tax disadvantage relative to the partnership form from 1987 to 1992. List and explain the factors that caused this outcome. Why...
-
For higher bracket taxpayers but not the highest their regular income tax rate might go up to about 28. However, the tax law will usually only tax their dividend income at only 15 Why? Do you agree...
-
In a balanced three-phase system, the abc-phase-sequence source is delta connected and Vab = 120 <30o V rms. The load consists of two balanced wyes with phase impedances of 10 + j1 and 20 + j5 . If...
-
What are the advantages and disadvantages of private warehousing?
-
Why did Congress pass the Health Care Quality Improvement Act of 1986?
-
Consider the following characteristics of either a JIT production system or a traditional production system. a. Products are produced in large batches. b. Large stocks of finished goods protect...
-
Required Information [The following Information applies to the questions displayed below.] Simon Company's year-end balance sheets follow. At December 31 Assets Current Year 1 Year Ago 2 Years Ago...
-
At 22 C and 1.0 atm, the enthalpy of solution of nitrogen in water is -11.0 kJ/mol, and its solubility is 6.68 10 -4 m. State whether the solubility of nitrogen is greater or less than 6.68 104 m...
-
The solubilities of most gases in water decrease as the temperature increases. Are the enthalpies of solution for such gases negative or positive? Explain your answer.
-
Explain why the following firms might choose a dual distribution strategy: a. Netflix b. Home Shopping Network c. Kohls
-
Westco Company issued 15-year bonds a year ago at a coupon rate of 8.1 percent. The bonds make semiannual payments and have a par value of $1,000. If the YTM on these bonds is 6.4 percent, what is...
-
Sexton Corporation has projected the following sales for the coming year. Sales 01 $720 Q2 $ 840 Q3 $ 960 04 $ 930 Sales in the year following this one are projected to be 10 percent greater in each...
-
Read the following news article about Hyatt Hotels: https://www.chicagotribune.com/business/ct-xpm-2014-02-14-chi-hyatt-earnings-20140214-story.html 1. What does this imply about the price elasticity...
-
Chapter 2 Organizing and Planning for Marketing Strategy 1 share your thoughts on the differences in role structures of a Medium to Large Company versus a Small Company 2 Research a company in which...
-
Be sure to answer ALL questions and ALL parts of the question. Credit only for correct and complete answers. For Christmas, Billy got a cute new pet, a Mogwai, for Christmas from his father while he...
-
You are in charge of analyzing five new product ideas and have been given the information shown in Table (1 = worst, 10 = best). Management has decided that criteria 2 and 3 are equally important and...
-
The MIT Sloan School of Management is one of the leading business schools in the U.S. The following table contains the tuition data for the masters program in the Sloan School of Management. a. Use...
-
Determine the force in each member of the roof truss. State if the members are in tension or compression.Assume B is a pin and C is a roller support. H60. 60 60 30 60 B 2 m - 2 m- 2 kN 2 kN
-
Determine the force in members JK, JN, and CD. State if the members are in tension or compression. Identify all the zero-force members. K 20 ft DE B - 20 ft- 30 ft- - 20 ft 2 k 2k
-
Determine the force in members GF, FC, and CD of the bridge truss. State if the members are in tension or compression. Assume all members are pin connected. 15 ft 30 ft B. - 40 ft-40 - 40 ft- 40 ft-...
-
2 . I have 2 gold coins, each 1 oz and 18K. I melt them to make a gold chain for myself. What is the purity of the gold chain? What is the weight of the gold chain?
-
jelle puts 1200 at the end of every year into Ira that averages 5% interest compounded annually. how much be be in the account when she retires after 35 years of investing
-
the Dalton family is purc a 155000 house .they plan to make 20% down payment. the ban will finance the balance for 30 years at 3.5% fixed rate calculate the amount of their payments and the total...

Study smarter with the SolutionInn App


