Identify two homonuclear diatomic molecules or ions with each of the following molecular orbital electron configurations. Are
Question:
Identify two homonuclear diatomic molecules or ions with each of the following molecular orbital electron configurations. Are these species stable?
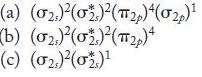
Transcribed Image Text:
(a) (02)(02)(2)(02) (b) (02)(0)(12)4 (c) (0)(0)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The image you provided seems to show three different molecular orbital MO electron configurations fo...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Identify two homonuclear diatomic molecules or ions with each of the following molecular orbital electron configurations. Are these species stable? (a) (0)(0)(2)(2p) (p) (0)(o)(2) 4 (02) (c) (0)(0)...
-
Coca-Cola Company and PepsiCo are two industry leaders in the carbonated soft drink industry. They are strong rivals of each other in the beverage market of the world. From 1975 to 1990s, both the...
-
Today in the spot market $1 = 1.82 Swiss francs and $1 = 130 Japanese yen. In the 90-day forward market, $1 = 1.84 Swiss francs and $1 = 127 Japanese yen. Assume that interest rate parity holds...
-
List all types of bonding present in the compound CaCO3. List all types of bonding present in the compound CaCO3. I. ionic bond II. polar covalent bond III. nonpolar covalent bond A. I only B. II...
-
Calculating Liquidity Ratios SDJ, Inc., has net working capital of $l,570 current liabilities of $4,380, and inventory of $1,875. What is the current ratio? What is the quick ratio?
-
As manager of Citywide Racquet Club, you must determine the best price to charge for locker rentals. Assume that the (marginal) cost of providing lockers is 0. The monthly demand for lockers is...
-
Explain the difference between requirements discovery and requirements gathering in general. Provide an example that clarifies the distinction between the two.
-
Tanaka Manufacturing Company is considering three new projects, each requiring an equipment investment of $22,000. Each project will last for 3 years and produce the following cash inflows. The...
-
How can organizations leverage ontologies and semantic technologies to enhance the discoverability and relevance of tacit knowledge embedded within unstructured data repositories? Explain
-
Use the molecular orbital diagram in Figure 10.40 to predict which species in each pair has the stronger bond. (a) F 2 or F 2 (b) O 2 or O + 2 (c) C 2+ 2 or C 2 Figure 10.40 Energy Atomic orbitals...
-
Identify the hybridization of the central atom that has the bonded-atom lone-pair arrangement of (a) A tetrahedron. (b) A trigonal bipyramid. (c) An octahedron.
-
Exercise 10.3 Consider a risky asset whose price $S_{t}$ is given by $$ \begin{equation*} d S_{t}=\sigma S_{t} d W_{t}+\frac{\sigma^{2}}{2} S_{t} d t \tag{10.25} \end{equation*} $$ where...
-
Performance and optimization assessment is important to identify performance degradation and bottleneck within a network. Recognizing the element to be measured may help to increase network...
-
1. if you've used any type of compute device, then you've interacted with an operating system (OS). Beyond a traditional desktop computer, identify one of these devices and describe its OS from a...
-
A new customer who wants to install Windows 8.1 on about 100 computers has contacted you. She tells you that the network uses Windows Server 2012 R2 in a domain configuration, and all new computers...
-
I need assistace in developing a scenario that will stimulate a discussion; "You are a security analyst working for an organization, and you have been tasked with implementing a certificate authority...
-
Task 1 Explore LAN design principles and their application in the network design process. 1.1. What do you mean by Network Design Model and explain the importance of having Network Design Models for...
-
A company announces that it will make a $1.00 dividend payment. Assuming all investors are subject to a 15 percent tax rate on dividends, how much should the companys share price drop on the...
-
TRUE OR FALSE: 1. Banks with a significantly large share of fixed-interest rate home loans are less exposed to interest rate risks. 2. Although Australian banks are pretty big, they are not...
-
The beam is made from three boards nailed together as shown. If the moment acting on the cross section is M = 600 N m, determine the resultant force the bending stress produces on the top board. 25...
-
If the built-up beam is subjected to an internal moment of M = 75 kN m, determine the maximum tensile and compressive stress acting in the beam. 150 mm 20 mm- 50 mm 150 mm 10 mm 10 mm 300 mm
-
If the built-up beam is subjected to an internal moment of M = 75 kN m, determine the amount of this internal moment resisted by plate A.
-
using Fiedler's Contingency Model discuss the leadership used by when changing Michael's performance. Provide your reasoning. Discuss why the coach's approach did not work based on this same theory...
-
10 points 2. Prepare the journal entry to record depreciation expense at the end of year 1, assuming the following: (If no entry is required for a transaction/event, select "No journal entry...
-
a. Create an aged accounts receivable according to your senior's instructions. Assume there are no receivables still outstanding prior to January 1, 2020. Note that the data set includes all orders...

Study smarter with the SolutionInn App


