Set up the equations needed to calculate the pH of a solution that is 0.050 M ammonium
Question:
Set up the equations needed to calculate the pH of a solution that is 0.050 M ammonium ion.
Strategy
The strategy is detailed in the steps listed in the margin.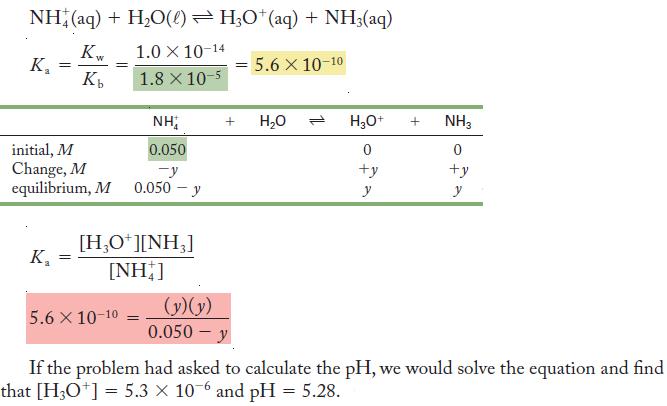
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
NHaq H2Ol H3O aq NH3aq K Kw Kb 10 x 1014 18 X 105 56 X 10...View the full answer

Answered By

Surojit Das
I have vast knowledge in the field of Mathematics, Business Management and Marketing. Besides, I have been teaching on the topics Management leadership, Business Administration, Human Resource Management, Business Communication, Accounting, Auditing, Organizer Behaviours, Business Writing, Essay Writing, Copy Writing, Blog Writing since 2020. It is my personality to act quickly in any emergency situations when students need my services. I am very professional and serious in every questions students asked me at the time of dealing any projects. I have been serving detailed, quality, properly analysed research paper through the years.
4.80+
91+ Reviews
279+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Calculate the pH of a solution that is 1.00 M HCN and 1.00 M HF. Compare the concentration (in molarity) of the CN2 ion in this solution with that in a 1.00 M HCN solution. Comment on the difference.
-
Calculate the hydronium ion concentration and pH of a solution that is 0.0500 M in HCl (a) Neglecting activities. (b) Using activities.
-
Calculate the pH of a solution that is (a) 0.0100 M in HClO4 and 0.0300 M in monochloroacetic acid. (b) 0.0100 M in HCl and 0.0150 M in H2SO4. (c) 0.0100 M in NaOH and 0.0300 M in Na2S. (d) 0.0100 M...
-
B. Giulia runs at 4.5 m/s (1 m/s is 2.2 mi/h ... just think "about 2 mi/h"). She is running eastward into the sunrise and along a straight-line path. Show a complete sketch from when you start...
-
Is a nucleus that absorbs at 6.50 more shielded or less shielded than a nucleus that absorbs at 3.20 ? Does the nucleus that absorbs at 6.50 require a stronger applied field or a weaker applied...
-
Would an employer ever want to increase the rate of employee turnover in a company? Why or why not?
-
DE19-9 Link Back to Chapter 5. Refer to Exhibit 19-9 on page 765. What would be Apex's beginning inventory if revenues, purchases and freight-in, and ending inventory are all the same as in Exhibit...
-
The balance sheet of Basket Rug Company reported the following: Requirements 1. Compute the book value per share for the common stock, assuming all preferred dividends are fully paid up (none in...
-
On November 30, Petrov Co. has $125.300 of accounts receivable and uses the perpetual inventory system Dec. 4 Sold $8,535 of merchandise (that had cost 55, 462) to customers on credit, teras 1/30. 9...
-
Calculate the pH of the following solutions. (a) 0.10 M sodium nitrate (b) 0.050 M KF Strategy Look at the ions formed when the salt dissolves. If both are spectator ions, the pH will be set by the...
-
Explain how to calculate the pH of the following solutions: (a) A solution that is 0.25 M KOH and 1.00 M ammonia (b) A solution that is 0.40 M HCl, 0.20 M HBr, 0.10 M HCOOH, and 0.20 M HF (c) A...
-
The following CVP income statements are available for Blanc Company and Noir Company. Instructions (a) Compute the break-even point in dollars and the margin of safety ratio (round to 3 places) for...
-
The Tokyo Olympics. After watching how the tokyo olympics became the most expensive summer game ever video answer the following questions. Q 3 : As you saw in the video, the capital investment a city...
-
write at least two paragraphs discussing the experiences of individuals who identify outside the traditional binary gender system (male/female.) Please explore the challenges they face and how...
-
Newly formed S&J Iron Corporation has 163,000 shares of $5 par common stock authorized. On March 1, Year 1, S&J Iron issued 9,000 shares of the stock for $12 per share. On May 2, the company issued...
-
Use the SMOKE for this question. The variable cigs is the number of cigarettes smoked per day. How many people in the sample do not smoke at all? What fraction of people claim to smoke 20 cigarettes...
-
Transcribed image text : Reproduced below from Farthington Supply's accounting records is the accounts receivable subledger along with selected general ledger accounts. Dec. 31/19 Balance Credit...
-
A car owner who knows no chemistry has to put antifreeze in his car's radiator. The instructions recommend a mixture of 30% ethylene glycol and 70% water. Thinking he will improve his protection he...
-
2. Assume a person bends forward to lift a load "with his back" as shown in Figure P12.42a. The person's spine piv- ots mainly at the fifth lumbar vertebra, with the principal supporting force...
-
The following is the expression for the vector E-field of an electromagnetic wave traveling in a homogeneous dielectric: Here v = 1.80 Ã 10 15 rad/s and k = 1.20 Ã 10 7 rad/m. (a)...
-
A tungsten lightbulb puts out 20 W of radiant energy (most of it IR). Assume it to be a point source and calculate the irradiance 1.00m away.
-
Consider an electromagnetic wave impinging on an electron. It is easy to show kinematically that the average value of the time rate-of-change of the electrons momentum vector p is proportional to the...
-
Given that rJ = 6.3%, rRF = 4.1%, and rM = 9.4%, determine the beta coefficient for Stock J that is consistent with equilibrium.
-
Simon Companys year-end balance sheets follow. At December 31 2017 2016 2015 Assets Cash $ 33,019 $ 37,839 $ 38,623 Accounts receivable, net 93,822 65,556 54,152 Merchandise inventory 117,963 89,253...
-
PLEASE REFER TO THE 2018 ANNUAL REPORT OF STARBUKS FOR THE YEAR FISCAL YR 2018, ENDING SEPTEMBER 30, 2018. Refer to the management discussion & analysis section and write a one page summary...

Study smarter with the SolutionInn App


