Toxic nitrogen monoxide gas can be prepared in the laboratory by carefully mixing a dilute sulfuric acid
Question:
Toxic nitrogen monoxide gas can be prepared in the laboratory by carefully mixing a dilute sulfuric acid with an aqueous solution of sodium nitrite, as the following equation shows. What volume of 1.22 M sulfuric acid (assume excess sodium nitrite) is needed to prepare 2.44 g NO?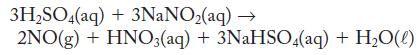
Transcribed Image Text:
3H₂SO4(aq) + 3NaNO₂(aq) → 2NO(g) + HNO3(aq) + 3NaHSO4(aq) + H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Deepak Pal
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
Refer to the Integrative Example on page 140. If 138 g Na 2 CO 3 in 1.42 L of aqueous solution is treated with an excess of NO(g) and O 2 (g), what is the molarity of the NaNO 2 (aq) solution that...
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
For this project, you must select an employer organization and research the organizations employee benefits package (plan). After you research the organizations employee benefits package, collect...
-
Plot the payoff diagrams fur the following instruments: (a) A caplet with cap rate Rcap = 6.75% written on 3-wonth Libor Lt, that is about to expire. (b) A forward contract written on a default-free...
-
Determine the derivative dP/dT of the function P = a ln T + bT 1 .
-
If \(10 \mathrm{~J}\) of magnetic potential energy is stored in a \(5.0-\mathrm{H}\) inductor, what is the current in the inductor?
-
The owner of a Major League Baseball team is considering moving his team from its current city in the upper Midwest to a city in the Southeast that offers a larger television market, a new stadium,...
-
26 The number of protons, neutrons, electrons in some particles are shown in the table below Particle Protons Neutrons electrons P 1 1 2 Q 2 2 2 R 3 4 2 T 4 5 4 Which one of the following particles...
-
1:- As a result of MEXIT, Telford Engineering had lost 30% of its pre-MEXIT export sales to CETA customers, due to increased trade and tariff barriers with CETA. (See P/L account before MEXIT in the...
-
Although silver chloride is insoluble in water, adding ammonia to a mixture of water and silver chloride causes the silver ions to dissolve because of the formation of [Ag(NH 3 ) 2 ]+ ions. What is...
-
Sodium thiosulfate, Na 2 S 2 O 3 , is used in photographic film developing. Th e amount of Na 2 S 2 O 3 in a solution can be determined by a titration with I 2 , according to the following equation:...
-
The purpose of producing 99 Mo (usually by neutron activation of natural molybdenum, as in the preceding problem) is to produce 99m Tc. Using the rules, verify that the - decay of 99 Mo produces 99m...
-
Compile the work you have done throughout this class for the department of the government that you chose: Create two deliverables: one for your contact at the department, and one for your professor....
-
CTL (Concrete Testing Lab) borrowed $80,000 for new equipment at 8 percent per year, compounded quarterly. It is to be paid back over 3 years in equal quarterly payments. For each part below, use...
-
Joseph Grant's will provided that $1 million was to be placed in trust for his son, Steven, for life, with a remainder to Stevens children per stirpes. What are the estate tax consequences to...
-
Prove that if \(X\) is \(d\)-dimensional standard normal, then \[ \lim _{|x|_{d} ightarrow \infty} \frac{E|x-X|_{d}}{|x|_{d}} ightarrow 1 \]
-
Thirty companies comprise the DJIA. Just how big are these companies? One common method for measuring the size of a company is to use its market capitalization, which is computed by multiplying the...
-
What is the effect on net income of not recording sales returns?
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
Draw the structure of each of the following compounds. a. (R) -2-Ethoxy-1, 1-dimethylcyclobutane b. Cyclopropyl isopropyl ether
-
Show that the van der Waals and RedlichKwong equations of state reduce to the ideal gas law in the limit of low gas density.
-
An initial step in the biosynthesis of glucose C 6 H 12 O 6 is the carboxylation of pyruvic acid CH 3 COCOOH to form oxaloacetic acid HOOCCOCH2COOH CH 3 COCOOH(s) + CO 2 (g) HOOCCOCH 2 COOH(s) If...
-
Fisk Corporation is trying to improve its inventory control system and has installed an online computer at its retail stores. Fisk anticipates sales of 38,400 units per year, an ordering cost of $2...
-
Modify the __init__ function take in two positional arguments material and summon. These are Metal and False by default. - make function called generate_probabilities which has two parameters -...
-
Using a 50%; 50% probabilities binary-interest-rate-tree model, and the following assumptions: r0 = 0.900%, rL = 2.200%, rH = 2.531%, volatility = 7%. Calculate the price of the 2-year 3.5%...

Study smarter with the SolutionInn App


