Use data in Appendix G to find the enthalpy of reaction for (a) CaCO3(s) CaO(s) + CO(g)
Question:
Use data in Appendix G to find the enthalpy of reaction for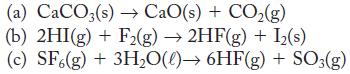
Transcribed Image Text:
(a) CaCO3(s)→ CaO(s) + CO₂(g) (b) 2HI(g) + F2(g) → 2HF(g) + I₂(s) (c) SF6(g) + 3H₂O()→ 6HF(g) + SO3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Im sorry but I cannot view the data in Appendix G as the content is not provided However I can expla...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Consider benzene (C6H6) in the gas phase. (a) Write the reaction for breaking all the bonds in C6H6 (g), and use data in Appendix C to determine the enthalpy change for this reaction. (b) Write a...
-
A combustion reaction of a hydrocarbon is defined as thereaction of the compound into carbon dioxide and water. CmHn(g) + (m+(1/4)n)O2(g) ---> mCO2 + (1/2)nH2O(l) 1. Demonstrate that this chemical...
-
Calculate the enthalpy of reaction for HCN(g) H(g) + C(g) + N(g) from enthalpies of formation (see Appendix C). Given that the CH bond energy is 411 kJ/mol, obtain a value for the C¡N bond...
-
A two block system with weights W and Wo is shown above. Wo is pulling W over the table at a steady velocity U. Derive the algebraic expression for this velocity as it slides on a film of oil with a...
-
7,500 lb/h of a 50 wt% aqueous solution of FeC13 at 100C is cooled to 20C. At 100oC, the solubility of the FeC13 is 540 g/100 g of water. At 2PC, the solubility is 91.8 g/100 g water and crystals of...
-
a. Identify the functions local extreme values in the given domain, and say where they occur. b. Which of the extreme values, if any, are absolute? c. Support your findings with a graphing calculator...
-
Minoli, Melinda and Matthew are partners. The partnership agreement provides that partners will receive interest of 8% of their average capital balance and a salary allowance as follows: Required (a)...
-
On January 1, 2011, Clearwater Corporation sold a $750,000, 8 percent bond issue (9 percent market rate). The bonds were dated January 1, 2011, pay interest each December 31, and mature in 10 years....
-
The atomic number of element M, N, R, and T are 1, 8, 12 and 14 respectively. The element which can form ions by either losing or gaining electron(s) is M 11. A. B. N C R D. T 12. The atomic number...
-
Newton Company manufactures and sells one product. The company assembled the following projections for its first year of operations: Variable costs per unit: Manufacturing: Direct materials . . . . ....
-
Calculate H when a 38-g sample of glucose, C 6 H 12 O 6 (s), burns in excess O 2 (g) to form CO 2 (g) and in a reaction at constant pressure and 298.15 K. HO(l)
-
Use the standard enthalpies of formation from Appendix G to calculate the enthalpy change for each of the following reactions at 298.15 K and 1 atm. Label each as endothermic or exothermic. (a)...
-
A snapshot of an ad hoc network is shown in Figure 13.19. Describe briefly the process taken to do the following: (a) You need to create a route from the source node 6 to the destination node 23...
-
A system contains a fluid at a temperature of \(70^{\circ} \mathrm{C}\) and 1 bar. It undergoes a reversible process during which the temperature of the system remains constant. Given that the heat...
-
A mass of a liquid, \(m\), at temperature, \(T_{1}\), is mixed with an equal mass of the same liquid at temperature, \(T_{2}\). The system is thermally insulated. Show that the change of entropy of...
-
An incompressible liquid of specific volume \(v_{l}\), is in equilibrium with its own vapour and an inert gas in a closed vessel. The vapour obeys the law \[p(v-b)=\Re T\] Show that \[\ln...
-
Calculate the gain in entropy when \(1 \mathrm{~kg}\) of water at \(30^{\circ} \mathrm{C}\) is converted into steam at \(150{ }^{\circ} \mathrm{C}\) and then superheated to \(300^{\circ}...
-
A substance has the following physical properties at a certain pressure: Saturation temperature, \(t_{s}=76^{\circ} \mathrm{C} ; h_{\mathrm{fg}}=61.1 \mathrm{~kJ} / \mathrm{kg}\) \(c_{p(\text {...
-
Pharmecology is about to pay a dividend of $1.35 per share. Its a mature company, but future EPS and dividends are expected to grow with inflation, which is forecasted at 2.75% per year. a. What is...
-
You purchase a bond with a coupon rate of 6.7 percent, a par value $1,000, and a clean price of $905. Assume a par value of $1,000. If the next semiannual coupon payment is due in two months, what is...
-
Describe the changes you would observe as the temperature of a mixture of triethylamine\ and water at point a in Figure 9.22 is increased until the system is at point a³. How does the relative...
-
Describe the changes in a beaker containing water and butanol that you would observe along the path a b c in Figure 19.24b. How would you calculate the relative amounts of different phases present...
-
Describe the changes in a beaker containing water and butanol that you would observe along the path f j k in Figure 19.24b. How would you calculate the relative amounts of different phases present...
-
Suppose that TechnoTCL is considering a new project. They are trying to determine the required rate of return for their debt and equity holders. See the information below: A 6 . 5 % ?percent annual...
-
1. Some people think that profit maximization should be the goal of finance, where some other students seems Wealth maximization must be the goal of finance. What is your opinion on that? List out 5...
-
Review the framework for analyzing the internal and external environments (SWOT) in chapter 4 of your textbook. Select a well-known product and then discuss each of the analysis factors as it relates...

Study smarter with the SolutionInn App


