Use the accompanying phase diagram to do the following: (a) Label each region of the diagram with
Question:
Use the accompanying phase diagram to do the following:
(a) Label each region of the diagram with the phase that is present.
(b) Identify the phase or phases present at each of the points A, B, C, D, and E.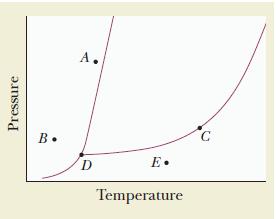
Transcribed Image Text:
Pressure B. A. D E. Temperature
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a The phases in each region are le...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
The volume of the rectangular solid above is 720. If AF = 15, which of the following is closest to the distance from C to F? 6 8 12 15 18 B F D 6 E
-
Use the accompanying phase diagram to do the following: (a) Label each region of the diagram with the phase that is present. (b) Identify the phase or phases present at each of the points G, H, J,...
-
Use the accompanying phase diagram for sulfur to answer the following questions. (The phase diagram is not to scale.) a. How many triple points are in the phase diagram? b. What phases are in...
-
Your friend recently attended a local mail fraud trial. In your conversation about the case, she described the cross-examination of the expert witness as follows: After his counsels questioning was...
-
A variable-frequency voltage source drives the network in figure. Determine the resonant frequency, Q, BW, and the average power dissipated by the network atresonance. w- 100 n 50 mH 12 cos wf V+ :5 F
-
Banner Mattress and Furniture Company wishes to study the number of credit applications received per day for the last 300 days. The sample information is reported below. Number of Credit Frequency...
-
What are the primary purposes of ESS and MSS? What are the advantages and disadvantages of each?
-
Ensign Landscape Design designs landscape plans and plants the material for clients. On April 1, there were three jobs in process, Jobs 39, 40, and 41. During April, two more jobs were started, Jobs...
-
An access matrix consists of 3 domains and 4 objects. 01 02 03 04 D1 OW orx orwc X D2 D3 rx rcw rcwcxc rcx rcx orw A process in domain D can grant a right R (r, w, or x) for object O to another...
-
The normal melting point of iodine is 113.5 C, and its normal boiling point is 184.3 C. The triple point occurs at 92.3 torr and 113.4 C, and the critical point is 512 C at 112 atm. Sketch the phase...
-
Answer the following questions by using the phase diagram in Exercise 11.44. (a) Sketch the heating curve that is expected when heat is added to the sample at constant pressure, starting at point J....
-
The use of cellular phones in automobiles has increased dramatically in the last few years. Of concern to traffic experts, as well as manufacturers of cellular phones, is the effect on accident...
-
Comparative financial statements for Weaver Company follow: Weaver Company Comparative Balance Sheet at December 31 This Year Last Year Assets Cash $ 6 $ 11 Accounts receivable 270 210 Inventory 105...
-
At what per annum rate must $260 be compounded daily for it to grow to $561 in 12 years? (Round to 100th of a percent and enter your answer as a percentage, e.g., 12.34 for 12.34%) (Assume 365 days...
-
During August, Diga Corporation plans to serve 44,000 customers. The company uses the following revenue and cost formulas in its budgeting, where q is the number of customers served: Revenue: $4.38q...
-
Consider the information below about the following three stocks: Amethyst, Sapphire and Tanzanite. Market Price Amethyst $26 Sapphire $16 Tanzanite $10 Number of Shares 4 20 3 Cash $10 $65 $22 Debt...
-
Entry for Uncollectible Accounts Easy Rider International is a wholesaler of motorcycle supplies. An aging of the company'saccounts receivableon December 31, 20Y3, and a historical analysis of the...
-
Continuous improvement recognizes that many small improvements add up to sizable benefits. Will continuous improvement take a company at the bottom of an industry to the industry to the top? Explain.
-
Refer to Exercise 8.S.I. Construct a scatterplot of the data. Does the appearance of the scatterplot indicate that the pairing was effective? Explain. Exercise 8.S.I. A volunteer working at an animal...
-
Calculate the density of oxygen gas at 37.4 C and a pressure of 720 mmHg.
-
A sample of gas has a mass of 0.205 g. Its volume is 0.112 L at a temperature of 25 C and a pressure of 740 mmHg. Find its molar mass.
-
A 1.50-L mixture of helium, neon, and argon has a total pressure of 754 mmHg at 310 K. If the partial pressure of helium is 431 mmHg and partial pressure of neon is 211 mmHg, what mass of argon is...
-
d) Calculate the expected return, in percent, for each asset assuming you buy the asset at the market price in t = 0 and you hold it until t = 2 when the final payoff is realized.
-
Current forecasts are for ABCCompany to pay dividends of $2, $2.44, and $5.30 over the next three years, respectively. At the end of three years you anticipate selling your stock at a market price of...
-
You are 22 years old, have saved up $40,000, and have a goal of being a millionaire by age 50.You intend to make monthly contributions into your investment account each month. You estimate you can...

Study smarter with the SolutionInn App


