Question: Write the expression for the equilibrium constant (K c ) for the following: (a) PC15(g) (b) 2NO2(g) (c) 2SO3(g) (d) H(g) + 12(g) 2HI(g) PC13(g)
Write the expression for the equilibrium constant (Kc) for the following: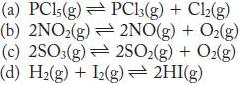
(a) PC15(g) (b) 2NO2(g) (c) 2SO3(g) (d) H(g) + 12(g) 2HI(g) PC13(g) + Cl(g) 2NO(g) + O(g) 2SO(g) + O(g)
Step by Step Solution
3.27 Rating (153 Votes )
There are 3 Steps involved in it
a K c K PC... View full answer

Get step-by-step solutions from verified subject matter experts


