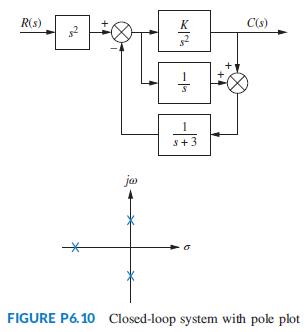
Find the value of K in the system of Figure P6.10 that will place the closed-loop poles
Question:
Find the value of K in the system of Figure P6.10 that will place the closed-loop poles as shown.
Transcribed Image Text:
R(s) K C(s) s+3 ja FIGURE P6.10 Closed-loop system with pole plot
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
To find the value of K we shoul...View the full answer

Answered By

MORINKE KUDAOS
I am an educator that has the necessary abilities and expertise owing to extensive interaction with students. I present answers to a variety of problems with step-by-step explanations, a well-thought-out strategy, and an easily understood breakdown. My objective is to teach students more easy methods and comprehension so that they may attain more success.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Find the value of k that makes f(x) = kx (5 - x), 0 x 5, a valid PDF.
-
Find the value of k that makes f(x) = kx2 (5 - x)2, 0 x 5, a valid PDF.
-
Find the value of k such that the system of linear equations is inconsistent. 1. 2. (4x 8y = -3 |2r + ky = 16 15 + 3D 6 10x + ky = 9
-
4. (23 pts) A plate is loaded so that a state of plane strain (Ezz- &zx=&zy =0) exists. (a) Determine the displacements for the plate for the deformations shown, i.e. finding u-Cixy and v=C2xy. (b)...
-
Suppose the production of airframes is characterized by a Cobb-Douglas production function: Q = LK. The marginal products for this production function are MPL = K and MPK = L. Suppose the price of...
-
Corporate warfare can get nasty. One firm may decide to acquire another. How does it do this? What can the target corporation do to prevent its being taken over? Should stockholders of either...
-
Pick a product like Cadillac or Sara Lee Deli products or service like Mr. Clean Performance Car Wash or a car and home insurance company that is offered in a limited number of countries. Assess the...
-
The following procedures were recently installed by Pine Creek Company: a. Along with petty cash expense receipts for postage, office supplies, etc., several post-dated employee checks are in the...
-
4. (50 points) The goal of this simulation study is to study the relative performance of uniform search, golden section search, and parabolic interpolation (algorithms 1-3 from class notes). Fix m...
-
The Candy Co. of Lethbridge pays its workers twice each month. Data for the two pay periods on June, are shown below: Union dues must be remitted to the union treasurer by the 15th of the following...
-
The closed-loop transfer function of a system is Determine the range of K 1 in order for the system to be stable. What is the relationship between K 1 and K 2 for stability? s + KIs+ K2 +K1s3 + K2s...
-
Find the range of K to keep the system shown in Figure P6.9 stable. R(s) + E(s) C(s) K S- 1 2+ 2s + 1 FIGURE P6.9
-
In the batch preparation of an aqueous solution, the water is first heated to 80C in a jacketed, agitated vessel; 1000 Imp. gal. (4545 kg) is heated from 15C. If the jacket area is 300 ft 2 (27.9 m 2...
-
Save Answer The frequency of shedding of vortices from a body owing to the flow of fluid past it depends on the length-scale of the body, the speed of the fluid flow, and the density and viscosity of...
-
12. The value of a product offering is determined by the customer and varies accord- ingly. How does a retailer like Walmart deliver value differently than Banana Republic? 13. Explain how Apple...
-
A box of height hand width w is joined along a plane 0. The joint strength has a yield stress, Oy. A force P is applied to the box shown in the diagram below. h = 20mm w = 30mm = 55 degrees P= 150kN...
-
Carbon dioxide levels in the Earth's atmosphere have risen from a pre-1800 baseline of 275 ppm to what average level? O About 500 parts per million CO. O About 400 parts per million CO. O About 600...
-
Practice: 0.100 mol of magnesium metal and a volume of aqueous hydrochloric acid that contains 0.500 mol of HCl are combined and react to completion. How many liters of hydrogen gas, measured at STP,...
-
Draw the pH-activity profile for an enzyme with one catalytic group at the active site. The catalytic group is a general-acid catalyst with a pKa of 5.6.
-
The words without recourse on an indorsement means the indorser is: a. not liable for any problems associated with the instrument. b. not liable if the instrument is dishonored. c. liable personally...
-
The following figure is an exploded view of a box. Identify and label all the components. TT T T T T
-
Draw a conceptual sketch of an incandescent light bulb. Identify all the components using arrows and numbers as in Figure 1.1. Figure 1.1 11 Radiator: Battery: 10 Spare Tire: Exhaust Manifold: 9. Gas...
-
There are 39 inches in a meter. What is the volume in the Engineering English system of a spherical apple that is 10. cm (note the decimal point here) in diameter? Assume that on the surface of the...
-
Name three major reports that a business owner should routinely access from QuickBooks. For each report, give three examples of the useful information that each can provide to a business owner or...
-
Discuss the key steps involved in entering vendor bills, recording expenses, and managing the accounts payable process within the program. Give three examples of bad things that might happen to the...
-
Trial Corporation is experiencing financial difficulties. One of its major customers, Jury Corporation, is concerned that if Trial is unable to continue in business, Jury's supply chain will be...

Study smarter with the SolutionInn App


