Four chemists are asked to determine the percentage of methyl alcohol in a certain chemical compound. Each
Question:
Four chemists are asked to determine the percentage of methyl alcohol in a certain chemical compound. Each chemist makes three determinations, and the results are the following:
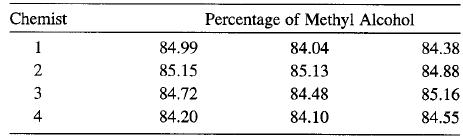
(a) Do chemists differ significantly? Use α = 0.05.
(b) Analyze the residuals from this experiment.
(c) If chemist 2 is a new employee, construct a meaningful set of orthogonal contrasts that might have been useful at the start of the experiment.
Transcribed Image Text:
Chemist Percentage of Methyl Alcohol 1 84.99 84.04 84.38 85.15 85.13 84.88 3 84.72 84.48 85.16 4 84.20 84.10 84.55
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
ANSWER To determine if the chemists differ significantly in their measurements of the percentage of methyl alcohol we can perform an analysis of variance ANOVA test a Hypothesis testing Null hypothesi...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Four laboratories (1-4) are randomly selected from a large population, and each is asked to make three determinations of the percentage of methyl alcohol in specimens of a compound taken from a...
-
You've been asked to determine the percentage of students who would support gay marriage. You want to take a random sample of fellow students to make the estimate. Explain whether each of the...
-
Social networking is becoming more and more popular around the world. Pew Research Center used a survey of adults in several countries to determine the percentage of adults who use social networking...
-
Consider a tank containing a liquid, and the rate of change of the liquid's height (h) with respect to time (t) is proportional to the difference between the current height and a reference height....
-
Sketch graphs of the following updating function over the given range and mark the equilibria. h(z) = e-z for 0 z 2.
-
First Firm's market cap is $5,940. This includes cash of $542. The firm has 299 shares outstanding. First Firm uses its cash to pay a dividend. If a retired lady has 61 shares, and needs a total of...
-
Gender in the Rock and Roll Hall of Fame From its founding through 2015, the Rock and Roll Hall of Fame has inducted 303 groups or individuals. Forty-seven of the inductees have been female or have...
-
The local residents of Greene County, a small, rural, mostly minority community, have recently learned that a major oil company is putting a refinery in the county. The residents ask for an...
-
Popular furniture company, IKEA, has purchased forests in Romania as well as land in Alabama to assist with keeping up with the wood demand necessary to complete customer orders. This was one way...
-
Explain the concept of hop count in RIP. Can you explain why no hop is counted between N1 and R1 in Figure 20.15? Figure 20.15 Hop counts in RIP NI N2 N3 N4 Source Destination RI R2 R3 1 hop (N4) 2...
-
The effective life of insulating fluids at an accelerated load of 35 kV is being studied. Test data have been obtained for four types of fluids. The results were as follows: (a) Is there any...
-
Three brands of batteries are under study. It is suspected that the lives (in weeks) of the three brands are different. Five batteries of each brand are tested with the following results: (a) Are the...
-
The survey asked respondents to classify their occupation. Can we infer that male heads (HHSEX: 1 = Male, 2 = Female) of households are more likely than their female counterparts to work as a manager...
-
Note that by the arbitrage-free principle, it is easy to show that when a stock pays a dividend D at t, the stock's value is immediately reduced by the amount of the dividend. In other words, liment,...
-
A large company with locations in different cities has taken an OOP approach in creating an administration program that manages all aspects of its business. These aspects include: the sale of all of...
-
b. If the query point q moves, the resulting q.3NN can change, and the point where q.3NN changes is called the safe exit point. Find the same exit point in the figure above when q moves in n2...
-
. Find YTM and Spot Rate using each Bond from A to C. Spot (ORN) (4) (5) (6) Period 1 2 3 YTM (1) (2) (3) Maturity 1 2 3 Based on your answers to Question 1, calculate prices of the following...
-
Translate the following E/R diagram to the relational model and write both relational schema and DDL queries for each relation. name Date surname Customer 1 orders Order age Status >18 & <100...
-
A student, starting from rest, slides down a water slide. On the way down, a kinetic frictional force (a nonconservative force) acts on her. The student has a mass of 83.0 kg, and the height of the...
-
The tractor is used to lift the 150-kg load B with the 24-mlong rope, boom, and pulley system. If the tractor travels to the right at a constant speed of 4 m/s, determine the tension in the rope when...
-
Write the charge balance for a solution of H 2 SO 4 in water if H 2 SO 4 ionizes to HSO 4 and SO 2 4 - .
-
For a 0.1 M aqueous solution of sodium acetate, Na + CH 3 CO - 2 , one mass balance is simply [Na + ] = 0.1 M. Write a mass balance involving acetate.
-
(a) Following the example of Mg(OH) 2 in Section 7-5, write the equations needed to find the solubility of Ca(OH) 2 . Include activity coefficients where appropriate. Equilibrium constants are in...
-
Do you look to social media to build your self-esteem? What aspects of social media make you feel better about yourself? What aspects make you feel worse? Do you think that social media has had an...
-
This is designed to help problem solvers speed up the slower process, slow other processes down to relieve pressure on the slow process, or a combination of the two. This statement BEST describes...
-
Write a routine request email to your supervisor asking for a 10% increase in your operating budget and to hire another team member for a new project your department is working on. The project is for...

Study smarter with the SolutionInn App


