Find the total resistance of each group of series resistors shown in Figure 467. Data in Figure
Question:
Find the total resistance of each group of series resistors shown in Figure 4–67.
Data in Figure 4–67 .
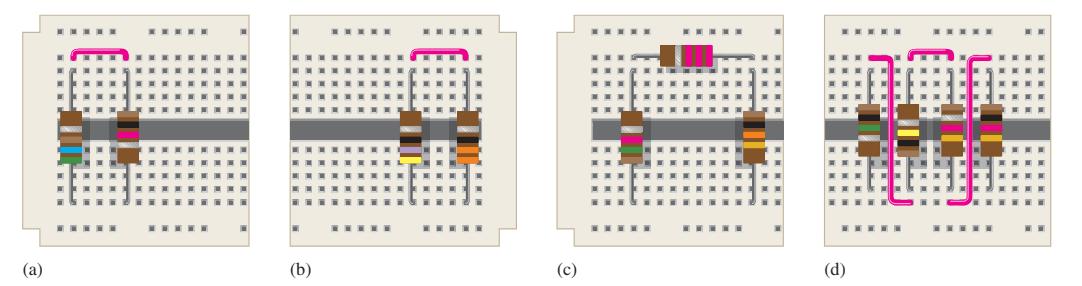
Transcribed Image Text:
(a) ***** (b) (c) **** (d) *****
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a R1R2 561G 102G 663G b R1R2 47G 33G 80G c R1R2...View the full answer

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The ages of employees at a company represent a. time series data b. quantitative data c. categorical data d. label data
-
In each of the following cases, decide whether or not a binomial distribution is an appropriate model, and give your reasons. a. A firm uses a computer-based training module to prepare 20 machinist...
-
One standard location for a pair of electrodes during an EKG is shown in the figure (Figure 1) . The potential difference ? V 31= V 3? V 1 is recorded. For each of the three instants a, b, and c...
-
Suppose that In Example 18.6 the electrical firm does not have enough prior information regarding the population mean length of life to be able to assume a normal distribution for p. The firm...
-
At the beginning of 2013, Eva Event Service had the following normal balances in its accounts: Account ............ Balance Cash .............. $60,000 Accounts Receivable ........ 38,000 Accounts...
-
Following are financial statements for Moore Company and Kirby Company for 2013: ¢ Moore purchased 90 percent of Kirby on January 1, 2012, for $657,000 in cash. On that date, the 10 percent...
-
You want to determine the average mercury content of the tuna fish consumed by U.S. residents. Suggest a sampling method that is likely to produce a representative sample. Explain why you chose this...
-
Following are a trial balance and the transactions that relate to the City of Patin Heights Debt Ser-vice Fund: 1. The city council of Patin Heights adopted the budget for the Debt Service Fund for...
-
A downtown bank promises its potential employees a $ 9000 sign-on bonus, an additional bonus of $ 13000 two years from now, an additional bonus of $ 16000 four years from now, and an additional bonus...
-
Connect each set of resistors in Figure 465 in series between points A and B. A o (a) R www R R3 www FIGURE 4-65 -O B R (b) R R3 B R www R4 R Ju ww R (c) R5 B
-
Determine the total power in each circuit of Figure 559. +0 (a) R 1.0 R 2.2 10 FIGURE 5-59 +0 R 1.0 (b) 10 mA R 2.2 R3 2.3.3 R4 5.6
-
What contracts are normally involved in establishing a business website?
-
Would you expect a portfolio that consisted of the NYSE stocks to be more or less risky than a portfolio of NASDAQ stocks?
-
Reasonable assurance means: a. the auditor disclaims responsibility for the audit opinion. b. an adverse audit report. c. the auditor has done adequate work to report whether or not anything came to...
-
What is a Dutch auction, and what company used this procedure for its IPO?
-
Explain, using examples, the following, based on what you read in the textbook under the heading ecological succession: a) The pioneer and climax stages. b) Ecological stability. c) Biodiversity and...
-
How does the use of financial leverage affect stockholders control position?
-
Lanthanum was reacted with hydrogen in a given experiment to produce the nonstoichiometric compound LaH2.90. Assuming that the compound contains H 2 , La 2+ , and La 3+ , calculate the fraction of La...
-
Find the radius of convergence of? 1.2.3 1.3.5 (2n-1) r2n+1 -1
-
Chemists and engineers must be able to predict the changes in chemical concentration in a reaction. A model used for many single-reactant processes is Rate of change of concentration = -kC n where C...
-
Chemists and engineers must be able to predict the changes in chemical concentration in a reaction. A model used for many singlereactant processes is Rate of change of concentration = -kC n where C...
-
The following list gives the measured gas mileage in miles per gallon for 22 cars of the same model. Plot the absolute frequency histogram and the relative frequency histogram. 23 25 26 25 27 25 24...
-
As a pharmacist describe how you would find these scenarios within the workplace completed by the pharmacist includes achievements, activities and also outcomes A) the pharmacist puts the customer at...
-
A $3600, 9.8% bond with semi-annual coupons redeemable at par in 4 years was purchased at 98.7. What is the average book value?
-
you will conduct a Micro-research project. You will make two original posts on the discussion board and two responses to your classmates during the unit. your 1st post: you will propose a simple...

Study smarter with the SolutionInn App


