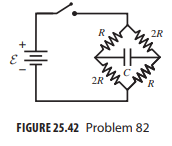
In the circuit of Fig. 25.42 the switch is initially open and the capacitor is uncharged. Find
Question:
In the circuit of Fig. 25.42 the switch is initially open and the capacitor is uncharged. Find expressions for the current I supplied by the battery (a) just after the switch is closed and (b) a long time after the switch is closed.
Transcribed Image Text:
R 2R 2R FIGURE 25.42 Problem 82
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Case1 Before switching ie t0 Capacitor is uncharged Switch Vo 0 V 0 Case2 to Just after closing the ...View the full answer

Answered By

Prafull Geed
About my education I have done my bachelor's degree in Electrical Engineering from Ujjain Engineering college, Ujjain with overall CGPA 0f 7.81/10. I am two times GATE qualified in Electrical Engineering in the year of 2020 and 2021. If I talk about my acedamics, I have secured 90.16% marks in 10th standard and 86.60% marks in 12th standard. About my tutoring experience, I used to teach my juniors, classmates, and my schoolmates.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The capacitor in the circuit of Fig. 16.38 is initially uncharged. Find v0 (t) for t > 0. 4i 262 2 580) V (+ 112
-
The capacitor in the circuit of Fig. 16.39 is initially uncharged. Find v 0 (t) for t > 0. 4i 9.68(t) V (+ 1F Vo
-
The switch in the circuit of Fig. 8.74 has been closed for a long time but is opened at t = 0 Determine i(t)for t > 0 . 12 V
-
The JoFe Computers and Accessory Company produces two types of laptop computer bags. Version A costs $32, takes 4 hours of labor, and sells for $50. Version B costs $38, takes 6 hours of labor, and...
-
Is a statement of cash flows really only a summary of cash receipts and disbursements recorded in the corporation's Cash account?
-
Your investment portfolio consists of $18,000 invested in only one stockMicrosoft. Suppose the risk-free rate is 6%, Microsoft stock has an expected return of 13% and a volatility of 44%, and the...
-
Bernardino Santos-Rodiguez was piloting a boat owned by his friend, Raul Viera-Torres, in waters near Guayama, Puerto Rico. The boat was equipped with a hydraulic steering system manufactured by...
-
AV City stocks and sells a particular brand of laptop. It costs the firm $625 each time it places an order with the manufacturer for the laptops. The cost of carrying one laptop in inventory for a...
-
8. A marine biologist measures the presence of a pollutant in an ocean and concludes that the concentration, C, in parts per million (ppm), as a function of the population, P, of the people who visit...
-
Phoenix Management helps rental property owners find renters and charges the owners one-half of the first months rent for this service. For August 2010, Phoenix expects to find renters for 100...
-
In Problem 60, take C 1 = C 2 = C, and find the current through R 2 as a function of time. Use the node and loop laws to get a differential equation for the current, and use the initial conditions on...
-
Stray voltage is a serious problem on dairy farms, often resulting from corroded wiring or poor wiring practices. These conditions can produce several volts between the ground and metal watering...
-
What are a few of the typical financing activities for a company like United Parcel Service, Inc. (UPS) , the world's largest package delivery company and a leading global provider of specialized...
-
Velation Electrons can be excited to any energy level and can drop to any lower energy level. When electrons drop from a higher energy level to energy level n=2, visible light is emitted. When...
-
"On my honor, as a Mississippi State University student, I have neither given nor received unauthorized assistance on this academic work." 10 Name: Preston Stroup You must show all of your work to...
-
E. A: LIOH, B: fructose, C: CaCO3 8. An analytical procedure requires a solution of chloride ions. How many grams of AICI, (Molar mass= 133g/mol) must be dissolved to make 2.00L of 0.150 M CI? A....
-
Based on the AHf data given, which compound is the most stable? HS(g), -20.6 kJ/mol NH4(g), +94.5 kJ/mol O PH3(g), +5.4 kJ/mol NH3(g), -46.4 kJ/mol NO4(g), +9.7 kJ/mol
-
16 Methanol is often used as an "antifreeze" for windshield washer fluid. How many moles of methanol would have to be added to 1000g of water to lower the freezing point of water to -20C? The...
-
Seven years ago, Halle (currently age 41) contributed $4,000 to a Roth IRA account. The current value of the Roth IRA is $9,000. In the current, year Halle withdraws $8,000 of the account balance to...
-
A seasonal index may be less than one, equal to one, or greater than one. Explain what each of these values would mean.
-
Using the differential form of G, dG = V dP S dT, show that if G mixing = nRT i x i ln x i , then H mixing = V mixing = 0.
-
Is a whale likely to get the bends when it dives deep into the ocean and resurfaces? Answer this question by considering the likelihood of a diver getting the bends if he or she dives and resurfaces...
-
Why is the preferred standard state for the solvent in an ideal dilute solution the Raoults law standard state? Why is the preferred standard state for the solute in an ideal dilute solution the...
-
What does Rafe represent to Elsa on the night they meet? Is it simply sex and loneliness? Or do you think there's something deeper involved? Another small defiance against her parents'...
-
Do all 6 questions. Each question is worth 10 marks, for a total of 60 available marks. The test is worth 40% of your course grade Question 1 ( 10 marks total) Elasticity, Competition versus...
-
Plant-based meat alternatives are on the rise as consumers become more health and environmentally conscious. Using the demand and supply analysis, how does this trend impact the meat industry in the...

Study smarter with the SolutionInn App


