The voltage across the 30k? resistor in Fig. 25.33 is measured with(a) a 50k? voltmeter,(b) a 250k?
Question:
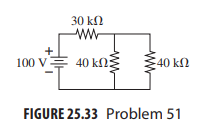
The voltage across the 30k? resistor in Fig. 25.33 is measured with(a) a 50k? voltmeter,(b) a 250k? voltmeter,(c) a 10M? digital meter. What does each read, to two significant figures?
Transcribed Image Text:
30 kΩ ww 100 V 40 kN 40 k2 FIGURE 25.33 Problem 51
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 20% (5 reviews)
Given R140k Resistor in parallel to a 40k resistor R240k Resistor in parallel to a 40k resistor and ...View the full answer

Answered By

CHRESTINE JEAN CASERES
I tutored Elementary and High School students in both English and Mathematics. I go to their house right after my class before when I was in college. Their house is 30 minutes ride from my house. I was able to teach them well because when I saw their report cards they are excelling and I am happy to see my students' progress.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A bowling ball with a radius of 15.0 cm travels down the lane so that its center of mass is moving at 3.60 m/s. The bowler estimates that it makes about 7.50 complete revolutions in 2.00 seconds. Is...
-
In Fig. 18.11b, the switch is closed at t = 0, and the capacitor begins to charge. What is the voltage across the resistor and across the capacitor, expressed as fractions of Vo (to two significant...
-
A voltmeter connected across a resistor always reads less than the actual voltage across the resistor when the meter is not present. Explain.
-
5. Explain how Culture can help companies set legal and ethical standards?
-
Assume the following income statement for the year ended December 31, 2017 and balance sheet at year-end: No equipment was purchased during the year. Equipment was sold for cash during the year....
-
Rally, Inc., is an all-equity firm with assets worth $22 billion and six billion shares outstanding. Rally plans to borrow $7 billion and use funds to repurchase shares. The firms corporate tax rate...
-
Lynn Goldsmith is a photographer known for her photographs of famous musicians. In 1981, Goldsmith had a photography session with the singer Prince. Three years later, Vanity Fair obtained a license...
-
Beginning inventory, purchases, and sales data for portable DVD players are as follows: June 1 Inventory ..... 75 units at $40 6 Sale .......... 60 units 14 Purchase ...... 90 units at $42 19 Sale...
-
On a private flight with capacity for 150 people, $800,000 per person is charged, plus $25,000 for each unsold seat on the plane. How many people must travel on the plane and what will be the price...
-
1. Sam's Menswear has $11,600 in Accounts Receivable at December 31. Sam's accountant estimates that $600 of the $11,600 will never be collected. Complete the current asset section of the balance...
-
Find all three currents in the circuit of Fig. 25.13 with the values given, but with battery ? 2 reversed. E = 6 V & = 9 V R1 = 20R3 = 10; R2 = 42 (a) Node A R Loop) R2 Loopa Node B Loop 3 (b) +
-
In Fig. 25.34, what are the meter readings when an ideal (a) Voltmeter (b) Ammeter is connected between A and B? 18 kN ww A 48 VE : 36 kn -B ww
-
George Stamper, a credit analyst with Micro-Encapsulators Corp. (MEC), needed to respond to an urgent e-mail request from the southeast sales office. The local sales manager reported that she had an...
-
O Macmillan Learning Solid ammonium chloride, NH Cl, is formed by the reaction of gaseous ammonia, NH3, and hydrogen chloride, HCl. NH3 (g) + HCl(g) NHCl(s) A 6.00 g sample of NH3 gas and a 6.00 g...
-
The Rydberg equation gives us the mathematical relationship between: A) an explanation for the continuous range of energy values associated with atomic spectra B) wavelength and frequency C) electron...
-
18. If 5.0 moles of calcium reacts with excess oxygen as shown, 2 Ca(s) + O2(g) 2CaO(s) AHxn= -1,270 kJ a. The reaction releases 6,350 kJ. b. The reaction absorbs 6,350 kJ. c. The reaction releases...
-
When aluminum is placed in concentrated hydrochloric acid, hydrogen gas is produced. 2 Al(s) + 6 HCl(aq) ->> 2 AICI, (aq) + 3 H(g) What volume of H(g) is produced when 7.20 g Al(s) reacts at STP?...
-
Draw a roadmap and write a plan to find the mass ( g ) of the products based on the given amount of reactants. 2 H 2 O 2 - > 2 H 2 O + O 2 given: H 2 O 2 = 4 9 . 4 mg
-
Alex is 31 years old and has lived in Los Alamos, New Mexico, for the last four years where he works at the Los Alamos National Laboratory (LANL). LANL provides employees with a 401(k) plan and for...
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
Carbon tetrachloride melts at 250. K. The vapor pressure of the liquid is 10,539 Pa at 290. K and 74,518 Pa at 340. K. The vapor pressure of the solid is 270. Pa at 232 K and 1092 Pa at 250. K. a....
-
In Equation (8.16), (dP/dT) vaporization was calculated by assuming that V gas m >> V liquid m . In this problem, you will test the validity of this approximation. For water at its normal boiling...
-
The densities of a given solid and liquid of molar mass 122.5 gmol 1 at its normal melting temperature of 427.15 K are 1075 and 1012 kgm 3 , respectively. If the pressure is increased to 120. bar,...
-
Suppose you want to move out of your apartment and buy a small house. Let s assume that your income is $ 3 9 , 6 0 0 a year, your student loans cost $ 2 5 0 per month, and your car payment is $ 3 2 5...
-
An investor purchased a corporate bond for $ 9 1 5 . The bond pays 3 . 7 5 % interest and matures this year. Calculate the current yield on the bond investment at the time of purchase. Determine the...
-
There's an old cliche that says you can't inspect quality into a product. Similarly, can assessment instruments like CEAI assess or induce innovation into organizations?" Someone wrote in regards to...
Freedom Of Information The Law The Practice And The Ideal 4th Edition - ISBN: 052171608X - Free Book

Study smarter with the SolutionInn App


