Whats the ammeter reading in Fig. 25.31? 20 20 6 V 20
Question:
What’s the ammeter reading in Fig. 25.31?
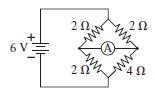
Transcribed Image Text:
20 20 6 V 20 Ω
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
In the above circuit to find current I apply ohms law V I R Where V Voltage V I Current A R Resistan...View the full answer

Answered By

AKILESH M
I has completed master degree in physics and very much interested in answering doubts related to physics. I can also teach students for better explanation of physics of concept this my strength too. I had scored 83% in masters degree and I hope the knowledge gained will be helpful for this work. I will do my work in the trusty manner.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) What current does the ammeter measure? (b) What should be the true current in the circuit (that is, the current without the ammeter present)? (c) By what percentage is the ammeter reading in...
-
(See Discussion Question Q25.14.) An ideal ammeter A is placed in a circuit with a battery and a light bulb as shown in Fig. Q25.15a, and the ammeter reading is noted. The circuit is then reconnected...
-
A galvanometer having a resistance of 25.0 has a 1.00 shunt resistance installed to convert it to an ammeter. It is then used to measure the current in a circuit consisting of a 15.0 resistor...
-
PC Connection is a leading mail order retailer of personal computers. A recent financial report issued by the company revealed the following information: Merchandise inventory (beginning of the...
-
Discuss the reasons why international business is much more complex today than it was 20 years ago.
-
We R Toys (WRT) is considering expanding into new geographic markets. The expansion will have the same business risk as WRTs existing assets. The expansion will require an initial investment of $45...
-
Mary Kay Morrow began working for Hallmark in 1982. At the beginning of 2002, Hallmark adopted the Hallmark Dispute Resolution Program, which required, among other things, that claims against the...
-
Nance Co. receives $280,000 when it issues a $280,000, 6%, mortgage note payable to finance the construction of a building at December 31, 2014. The terms provide for semiannual installment payments...
-
8. Two dice are rolled. Let A represent rolling a sum greater than 7. Let B represent rolling a sum that is a multiple of 3. Determine n(AB). A. 5 B. 8 C. 12 D. 15 Long Answer: Show all work for the...
-
Wagner Printers performs all types of printing, including custom work, such as advertising displays. and standard work. such as business cards. Market prices exist for standard work. and Wagner...
-
In Fig. 25.30, how much power is dissipated in the 4 resistor? 10 ww- 6 VE 29
-
In Fig. 25.32, find the equivalent resistance measured between A and B. A. R. 2R R 2R B- FIGURE 25.32 Problem 49
-
The financial statements of Marks and Spencer plc (M&S) are available at the books companion website or can be accessed at corporate.marksandspencer....
-
For normal construction involving field cast concrete, tack welding of reinforcing bar assemblies is permitted only when A. reinforcing bars are assembled off-site B. reinforcing bars are not...
-
Determine the number of moles present in 98.6 g of nitric acid.
-
If a chemical reaction has a positive AH and a positive AS, then A) it will be spontaneous at all temperatures. B) it will be non-spontaneous at all temperatures. C) it will be spontaneous at high...
-
How many calories would be needed to boil 2.5 gal of water that was at 24.0 degrees C to start with?
-
2.78 To which carbon atoms in a heptane molecule can each of the following alkyl groups be attached without extending the longest carbon chain beyond seven carbons? b. Isopropyl c. sec-Butyl a. Ethyl...
-
Compare and contrast the employer's responsibilities for providing a defined benefit plan to employees relative to providing a defined contribution plan.
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
For water, H vaporization is 40.656 kJmol 1 , and the normal boiling point is 373.12 K. Calculate the boiling point for water on the top of Mt. Everest of (elevation 8848 m), where the normal...
-
Calculate the difference in pressure across the liquidair interface for a (a) Mercury and (b) Methanol droplet of radius 125 nm.
-
Calculate the vapor pressure of CH 3 OH(l) at 298.15 K if He is added to the gas phase at a partial pressure of 200. Bar using the data tables. By what factor does the vapor pressure change?
-
An asset pays $ 2 0 0 annually forever starting next year ( t = 1 ) . The discount rate for the asset is 6 . 7 % . What is the value today ( t = 0 ) of the asset?
-
A company's bond has an annual coupon rate of 4 % . The bond pays coupons semiannually and has a par value of $ 1 , 0 0 0 . The bond matures in 6 years. What is the percentage ( % ) yield to maturity...
-
A bond pays a semi-annual coupon at an APR of 11.25%. The bond will mature in 6.00 years and has a face value of $1,000.00. The bond has a yield-to-maturity of 11.82% APR. What is the current yield...

Study smarter with the SolutionInn App


