Loomis, Inc. reported the following on the companys income statement in two recent years: a. Determine the
Question:
Loomis, Inc. reported the following on the company’s income statement in two recent years: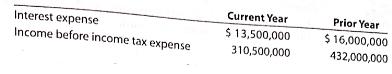 a. Determine the times interest earned ratio for the current year and the prior year. Round to one decimal place.
a. Determine the times interest earned ratio for the current year and the prior year. Round to one decimal place.
b. Is this ratio improving or declining?
Transcribed Image Text:
Current Year Prior Year Interest expense Income before income tax expense $ 13,500,000 $ 16,000,000 310,500,000 432,000,000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a We know Times Interest Earned EBIT Earning Before Interest and Taxes ...View the full answer

Answered By

Prashant Sharma
As I am DBF fro IUT , I have caliber to teach and full fill all academic needs like in teaching and training all the relevant subjects,preparations of assignments preparations of exams and any other problems related to educations, I give you very updated guidances and directions to our esteemed students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Accounting
ISBN: 9781337902687
28th Edition
Authors: Carl S. Warren, Christine Jonick, Jennifer Schneider
Question Posted:
Students also viewed these Business questions
-
Question 6 The government of Jonovia seizes all assets of Refresh, a world-famous, soft-drink brand in Jonovia, and offers compensation to the company. This action by the government of Jonovia is an...
-
The times interest earned ratio for Burlington Perfumes Inc. is 3.0, but the current ratio is only 1.2. Discuss future prospects for Burlington, given that the industry average for these ratios is...
-
* Times interest earned Loomis, Inc. reported the following on the companys income statement in two recent years: Current Year Prior Year Interest expense $499,000 $598,800 Income before income tax...
-
just finished the business plan of his start-up company. According to the projections he carried-out, the initial investment is 1,500,000 SAR (assume that we are in the beginning of 2023), which will...
-
Will a production quota in a competitive market always increase producer surplus?
-
The following accounting information pertains to Phoenix Co. and Roswell Co. companies at the end of 2017. The only difference between the two companies is that Phoenix Co. uses FIFO while Roswell...
-
In 2014, Barker contacted Price about a van Price had advertised for sale. The advertisement described the van as a 1994 Ford E350. Barker and Price agreed to meet, and, on April 9, Barker inspected...
-
Howies Carpet World has just received an order for carpets for a new office building. The order is for 4,000 yards of carpet 4 feet wide, 20,000 yards of carpet 9 feet wide, and 9,000 yards of carpet...
-
In the estimation of international trade's casual effect on country's income, InYa+BT+yWi+i, = where Y; denotes income per person, T; denotes international trade, W; denotes within-country trade and...
-
Prepare the Annual Reconciliation of Employer Wage Tax for Philadelphia, using the blank. For lines 1, 3, and 4, use gross wages and salaries per general ledger less exempt wages paid to Russell...
-
On the first day of the fiscal year, a company issues $35,000, 5%, five-year installment notes that have annual payments of $5,415. The first note payment consists of $1,750 of interest and $3,665 of...
-
On the first day of the fiscal year, a company issues $89,000, 6%, five-year installment notes that have annual payments of $21,128. The first note payment consists of $5,340 of interest and $15,788...
-
Evaluate this statement: If you sued someone, you would rather get nominal damages than punitive damages.
-
An experimental plot of In(k) vs. 1/T is obtained in lab for a reaction. The slope of the best-fit line for the graph is -1875 K. What is the value of the activation energy for the reaction in kJ/mol?
-
A 1 5 0 g sample of mercury and a 1 5 0 g sample of iron are at an initial temperature of 2 5 . 0 \ deg C . If 2 5 0 cal ( 1 0 5 0 J ) of heat is applied to each sample, what is the final temperature...
-
Given the equation 2H(g) + O (g) 2HO (g), is it correct to say that 10 g of hydrogen will react with 10 g of oxygen to produce 20 g of water vapor? False True
-
An irregularly shaped object has a density of 1 . 0 5 0 g / cm 3 and a mass of 1 2 . 4 g . How much water in mL will be displaced when the object is immersed into water? ( 1 cm 3 = 1 mL
-
Absolute zero is the theoretical coldest temperature in the universe. It is defined as 0 K . What is the value for absolute zero in \ deg C ?
-
One of the linkages in squalene is tail-to-tail, not head-to-tail. What does this suggest about how squalene is synthesized in nature?
-
What are technical skills At what level are they most important and why?
-
Wasserman Corporation issued 10-year bonds on January 1, 2011. Costs associated with the bond issuance were $160,000. Wasserman uses the straight-line method to amortize bond issue costs. Prepare the...
-
On January 1, 2011, Henderson Corporation retired $500,000 of bonds at 99. At the time of retirement, the unamortized premium was $15,000 and unamortized bond issue costs were $5,250. Prepare the...
-
Coldwell, Inc. issued a $100,000, 4-year, 10% notes at face value to Flint Hills Bank on January 1, 2011, and received $100,000 cash. The note requires annual interest payments each December 31....
-
Explain the observations below: CuCl N. Cu CUN N. N + 2+ OH2 HO OH2 HO OH2 OH2 Colorless red, molar absorptivity around 7200 M-1 cm-1 blue, molar absorptivity ~10 M-1 cm-1
-
12. Is the following unbalanced reaction thermodynamically favorable? Why or why not? 2+ 2+ +
-
1. What factors are most likely to influence an adolescent's response when experiencing peer pressure to experiment with alcohol or drugs? What role do parents play? 2. The book discusses the...

Study smarter with the SolutionInn App


