These financial statement items are for Norsted Company at year-end, July 31, 2022. Instruction a. Prepare an
Question:
These financial statement items are for Norsted Company at year-end, July 31, 2022.
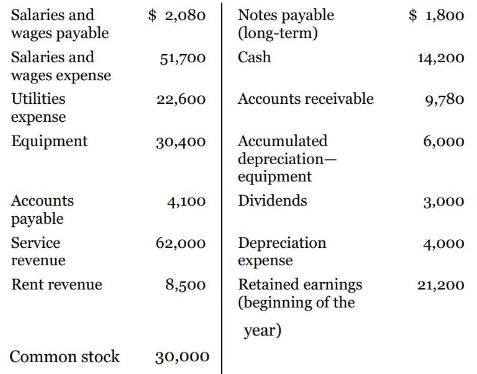
Instruction
a. Prepare an income statement and a retained earnings statement for the year.
b. Prepare a classified balance sheet at July 31.
Transcribed Image Text:
Salaries and $ 2,080 $ 1,800 Notes payable (long-term) wages payable Salaries and 51,700 Cash 14,200 wages expense Utilities 22,600 Accounts receivable 9,780 expense Equipment Accumulated depreciation- equipment 30,400 6,000 Accounts 4,100 Dividends 3,000 payable Service 62,000 Depreciation 4,000 revenue expense Retained earnings (beginning of the Rent revenue 8,500 21,200 year) Common stock 30,000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a NORSTED COMPANY Income Statement For the Year Ended July 31 2022 Revenues Service revenue 62000 Re...View the full answer

Answered By

Ayush Jain
Subjects in which i am expert:
Computer Science :All subjects (Eg. Networking,Database ,Operating System,Information Security,)
Programming : C. C++, Python, Java, Machine Learning,Php
Android App Development, Xamarin, VS app development
Essay Writing
Research Paper
History, Management Subjects
Mathematics :Till Graduate Level
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Financial Accounting
ISBN: 9781119594598
11th Edition
Authors: Jerry J. Weygandt, Paul D. Kimmel, Donald E. Kieso
Question Posted:
Students also viewed these Business questions
-
These financial statement items are for B. Snyder Company Inc. at year-end, July 31, 2011. Instructions (a) Prepare an income statement and a retained earnings statement for the year. (b) Prepare a...
-
These financial statement items are for B. Snyder Company at year-end, July 31, 2010. Instructions(a) Prepare an income statement and an owner??s equity statement for the year. The owner did not make...
-
These financial statement items are for Basten Company at year end, July 31, 2017. Instructions (a) Prepare an income statement and a statement of owner's equity for the year. The owner did not make...
-
Marks 1. Find the limits, if they exist. If a limit does not exist, check whether the function approaches +00 x2 + 2x - 15 (5) (a) lim x-3 x2-4x +3 x2 - 4 (5) (b) lim x2 x4 - 16 Carol Ferland CF...
-
Of each dollar spent on textbooks at college bookstores, 22.3 cents goes to the college store for profit, store operations, and personnel. On average, a college student at a four-year college spends...
-
Consider a system of C components with internal energy E(S, V, N, N2,..., NC). Find the differential and integrated forms of the fundamental equation for e = E/N, where N Ni. =
-
Explain events and their importance in dynamic modeling.
-
The following is an excerpt from a conversation between the chief executive officer, Rob Rameriz, and the chief financial officer, Maurice Chandler, of Nile Group, Inc.: Rameriz (CEO): Maurice, as...
-
2. Consider a discrete-time linear time invariant system with impulse response -38[n+1]+8[n] + 28[n-1] h[n] == a) Plot h[n]. Determine if this system is causal and stable. b) Find and plot the system...
-
While snoozing at the controls of his Pepper Six airplane, Dunse P. Sluggard leaned heavily against the door; suddenly, the door flew open and a startled Dunse tumbled out. As he parachuted to the...
-
Using Apples annual report, determine its current liabilities at September 30, 2017, and September 29, 2018. Were current liabilities higher or lower than current assets in these two years?
-
The adjusted trial balance for Martell Bowling Alley at December 31, 2022, contains the following accounts. Instructions a. Prepare a classified balance sheet; assume that $22,000 of the note payable...
-
A proton is launched from point 1 in Figure Q21.20 with an initial velocity of \(3.9 \times 10^{5} \mathrm{~m} / \mathrm{s}\). By how much has its kinetic energy changed, in \(\mathrm{eV}\), by the...
-
A 69-g ice cube at 0C is placed in 981 g of water at 30C. What is the final temperature of the mixture? C Additional Materials eBook
-
An electron, which is originally at rest, is accelerated by a constant electric field of 220 N/C between two oppositely charged plates. If the plates are 0.42 mm apart, what speed will the electron...
-
design and code several class templates and test them on different instantiations. Learning Outcomes Upon successful completion of this workshop, you will have demonstrated the abilities to: design...
-
A rocket engine can accelerate a rocket launched from rest vertically up with an acceleration of 23.4 m/s. However, after 50.0 s of flight the engine fails. Ignore air resistance. What is the...
-
Write a Java Servlet Program that represents a pseudo ATM machine where the user will authenticate their userID from the client-side and after successful authentication make a deposit, withdraw funds...
-
Austin Grocers recently reported the following 2012 income statement (in millions of dollars): Sales...................................................................................700 Operating...
-
Compare and contrast debt financing and equity financing as ways of starting a new business. Does one have an overall advantage over the other? What situation is more favorable to the use of debt...
-
(a) What are the major sources (inflows) of cash in a statement of cash flows? (b) What are the major uses (outflows) of cash?
-
(a) What are the major sources (inflows) of cash in a statement of cash flows? (b) What are the major uses (outflows) of cash?
-
Why is it important to disclose certain noncash transactions? How should they be disclosed?
-
Given the matrix A -3 0 -14 3 4 6 9115 7701 a) Determine all solutions of the homogeneous system Ax = 0. b) Determine if the columns of A span R.
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
On January 1, 2023, Holland Corporation paid $7 per share to a group of Zeeland Corporation shareholders to acquire 60,000 shares of Zeeland's outstanding voting stock, representing a 60 percent...

Study smarter with the SolutionInn App


