The solubility products for a series of hydroxides are Which hydroxide has (a) The lowest molar solubility
Question:
The solubility products for a series of hydroxides are
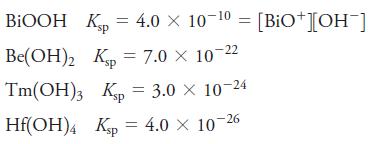
Which hydroxide has
(a) The lowest molar solubility in H2O?
(b) The lowest molar solubility in a solution that is 0.35 M in NaOH?
Transcribed Image Text:
BIOOH Kp 4.0 10- = = Be(OH)2 Kp = 7.0 10-2 Tm(OH)3 Kp = 3.0 10-24 sp Hf(OH)4 Kp = 4.0 10-26 [BIO+][OH-]
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a solubility in water is calculated as 1 BiOOH Ksp BiO OH 4010 10 s ...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
The solubility products for a series of iodides are CuI K = 1 x 10-12 AgI Ksp = 8.3 x 10-17 PbI2 Ksp = 7.1 x 10-9 BiI3 Ksp = 8.1 x 10-19 List these four compounds in order of decreasing molar...
-
Is it possible for a series of positive terms to converge conditionally? Explain.
-
From the solubility data given, calculate the solubility products for the following compounds: (a) SrF2,
-
Stock X has 32% standard deviation of return. Stock Y has 32% standard deviation of return. The correlation between the returns of the two stocks is 64%. Buck's portfolio consists of equal...
-
This problem investigates the sensitivity of the prices of bonds carrying differing coupon rates to interest rate changes. Bonds K and L both have a face value of $1000 and 15 years remaining until...
-
Suppose U.S. aggregate output is still below potential by 2018, when a new Fed chair is appointed. Suppose his or her approach to monetary policy can be summarized by the following statement: I care...
-
A surface condenser deals with \(13625 \mathrm{~kg}\) of steam per hour at a pressure of \(0.09 \mathrm{bar}\). The steam enters 0.85 dry and the temperature at the condensate and air extraction...
-
According to a summary of the payroll of Apline Publishing Co., $460,000 was subject to the 7.5% FICA tax. Also, $39,000 was subject to state and federal unemployment taxes. a. Calculate the...
-
Discuss the significance of inherent safety principles within PSM. How can process design and operational strategies be optimized to reduce inherent risks and promote safety throughout the lifecycle...
-
The horizontal rigid beam ABCD is supported by vertical bars BE and CF and is loaded by vertical forces P1 = 400 kN and P2 = 360 kN acting at points A and D, respectively (see figure). Bars BE and CF...
-
Calculate the solubility-product constant for each of the following substances, given that the molar concentrations of their saturated solutions are as indicated: (a) AgSeCN (2.0 x 10 -8 M; products...
-
Copper was determined in a river water sample by atomic absorption spectrometry and the method of standard additions. For the addition, 100.0 L of a 1000.0-g/mL Cu standard was added to 100.0 mL of...
-
Suppose that in April 2019 Van Dyck Exponents offered 100 shares for sale in an IPO. Half of the shares were sold by the company and the other half by existing shareholders, each of whom sold exactly...
-
A sports memorabilia collector propositions a sport journalist with access to the locker rooms during Super Bowl LI to infiltrate the New England Patriots locker room and take Tom Brady's jersey in...
-
What type of communication strategies are predominant? Considering the many available mediums, is TV advertising still the most powerful, or has it faded in importance?
-
What is ethical? what is right? What is right for the world of work, as you are shaping it in your professional practice?
-
How does motivation affect our survival as humans? Consider in your answer the role of dopamine in motivated behaviors and the different types of stimuli that we are motivated to seek out. Choose...
-
A 50 Kg toolbox is being push along the floor by a 100 N horizontal force. Friction between the floor surface and the box is 20 N. What is the acceleration of the box?
-
Do you believe technical evangelism effectively socializes Infosys's employees to accept the company's culture ideals?
-
Three successive resonance frequencies in an organ pipe are 1310, 1834, and 2358 Hz. (a) Is the pipe closed at one end or open at both ends? (b) What is the fundamental frequency? (c) What is the...
-
Distinguish between (a) Voltammetry and amperometry. (b) differential-pulse voltammetry and square-wave voltammetry. (c) A limiting current and a diffusion current. (d) The standard electrode...
-
It has been suggested that many polarograms can be obtained on a solution without depleting the electroactive analyte. Suppose that in a polarographic experiment we monitor the limiting current for...
-
An unknown cadmium(II) solution was analyzed polarographically by the method of standard additions. A 25.00-mL sample of the unknown solution produced a diffusion current of 1.86 (A. Following...
-
In Unix and Linux systems, the configuration of the operating system, servers, applications and preferences is commonly stored in plain text files. You want to view the hardware attached to the...
-
An actual corporation reported this information on their 2019 Annual Report ($ millions). Inventory cost method of LIFO is used. Date Inventory Balance Cost of Goods Sold (COGS) December 31, 2018...
-
The markets in general are paying a 2% real rate of return. Inflation is expected to be 3%. ABC stock commands a 6% risk premium. What is the expected rate of return on ABC stock? The required rate...

Study smarter with the SolutionInn App


