Titration of 50.00 mL of 0.04715 M Na 2 C 2 O 4 required 39.25 mL of
Question:
Titration of 50.00 mL of 0.04715 M Na2C2O4 required 39.25 mL of a potassium permanganate solution.
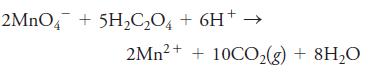
Calculate the molar concentration of the KMnO4 solution.
Transcribed Image Text:
2MNO4 + 5H2C,O, + 6H+ 2Mn?+ + 10CO,(g) + 8H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
For this redox titration we use the formu...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
Calculate the molar concentration of a solution that is 50.0% NaOH (w/w) and has a specific gravity of 1.52.
-
Calculate the molar concentration of a 20.0% solution (w/w) of KCl that has a specific gravity of 1.13
-
Calculate the molar concentration of a dilute Ba(OH)2 solution if (a) 50.00 mL yielded 0.1791 g of BaSO4. (b) Titration of 0.4512 g of primary standard potassium hydrogen phthalate (KHP) required...
-
The pilot of a small boat charts a course such that the boat will always be equidistant from an upcoming rock and the shoreline. Describe the path of the boat. If the rock is 2 miles offshore, write...
-
Using the bond yield given in the final column of Table 15.2, verify the September 22, 2009, quoted price for the Province of New Brunswick 4.4% coupon bond, maturing June 3, 2019.
-
Kensington Company uses special journals. It recorded in a sales journal a sale made on account to R. Stiner for $435. A few days later, R. Stiner returns $70 worth of merchandise for credit. Where...
-
Explain the importance of a written contract.
-
Pratt Corp. started the Year 2 accounting period with total assets of $30,000 cash, $12,000 of liabilities, and $5,000 of retained earnings. During the Year 2 accounting period, the Retained Earnings...
-
Draw the Logic Diagram for the following Boolean expressions. a) F = (x'y + y'z' )(x+ y') (b) F A+BZ+A'B'Z'
-
Jay Rexford, president of Photo Artistry Company, was just concluding a budget meeting with his senior staff. It was November of 20x4, and the group was discussing preparation of the firms master...
-
The phosphorus in a 0.3019-g sample was precipitated as the slightly soluble (NH 4 ) 3 PO 4 . 12MoO 3 . This precipitate was filtered, washed, and then redissolved in acid. Treatment of the resulting...
-
A 0.7891-g sample of a mixture consisting solely of sodium bromide and potassium bromide yields 1.2895 g of silver bromide. What are the percentages of the two salts in the sample?
-
Now that you have read the chapter on investment fundamentals, what do you recommend to Shavenellyee and Sarena on the subject regarding? 1. Portfolio diversification for Shavenellyee? 2. Dollar-cost...
-
Write the expression in the form \(D \sin (\omega t+\phi)\). \(\cos t+\frac{1}{3} \sin t\)
-
Express each complex number in its polar form. \(-\frac{1}{2} j\)
-
Using real-world examples, discuss the possible relationship between economic growth and economic development.
-
Find all possible values for each expression. \(\sqrt{1+j \sqrt{2}}\)
-
Write the expression in the form \(D \sin (\omega t+\phi)\). \(\frac{1}{2} \cos 3 t-\sin 3 t\)
-
A transverse wave on a string is described by y(x, t) = (1.2 cm) sin [(0.50p rad/s)t (1.00p rad/m)x] Find the maximum velocity and the maximum acceleration of a point on the string. Plot graphs for...
-
Making use of the tables of atomic masses, find the velocity with which the products of the reaction B10 (n, ) Li7 come apart; the reaction proceeds via interaction of very slow neutrons with...
-
Indicate whether an aqueous solution of the following compounds is acidic, neutral, or basic. Explain your answer. *(a) NH4OAc (b) NaNO2 *(c) NaNO3 (d) NaHC2O4 *(e) Na2C2O4 (f) Na2HPO4 *(g) NaH2PO4...
-
Suggest an indicator that could be used to provide an end point for the titration of the first proton in H3AsO4.
-
Suggest a method for determining the amounts of H3PO4 and NaH2PO4 in an aqueous solution.
-
Recently, there has been a resurgence in labor union organization and a subsequent increase in union membership across the United States. What can today's labor unions learn from their past 140-year...
-
). Prove by contraposition: "Vm, ne Z, if m -n is even, then m - n is even (15 points)
-
The production function is Y = AKL-a (1) where L is labor at time t, K, is capital at time t, A, is TFP at time t and a (0, 1). The law of motion for capital is K = 8Y-8K Assume that TFP evolves...

Study smarter with the SolutionInn App


