Name this lipid. H3C(CH2)5CH ||| CH(CH)7 -0 CH -CH || -(CH2)12CH3 0 COO CH-O-P-0-CH-CH NH
Question:
Name this lipid.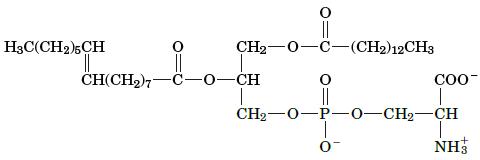
Transcribed Image Text:
H3C(CH2)5CH ||| CH(CH)7 -0 CH -CH || -(CH2)12CH3 0 COO CH-O-P-0-CH-CH NH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Phospholipid Phospholipids are a class of lip...View the full answer

Answered By

Hamza Amjad
Currently I am student in master degree program.from last two year I am tutring in Academy and I tought many O/A level student in home tution.
4.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Biochemistry Life At The Molecular Level
ISBN: 9781118918401
5th Edition
Authors: Donald Voet, Judith G Voet, Charlotte W Pratt
Question Posted:
Students also viewed these Sciences questions
-
Benedicts Test for Reducing Sugars: Tube Solution/food Color reaction to Benedicts solution Presence of reducing sugar (green/reddish orange color) 1 1ml distilled water 2 good pinch glucose powder 3...
-
1) In a bullet/ outline form, simply name the component (RNA, protein, membrane, etc.) used, ideally in the order used, and where appropriate add a brief descriptor of that component this should take...
-
A wood beam AB on simple supports with span length equal to 10 ft is subjected to a uniform load of intensity 125 lb/ft acting along the entire length of the beam, a concentrated load of magnitude...
-
Taylors 2022 health insurance premiums of $7,800 are paid by her employer. During 2022, Taylor requires surgery on her vocal chords. The cost of the surgery is $10,000 and Taylors insurance covers...
-
Describe three ways in which PS can be used to protect against DDos attacks.
-
Using Table 8.4, estimate ÎH for the following gas-phase reactions: a. b. c. Br Br Br Br TABLE 8.4 Single Bonds e Average Bond Enthalpies (kJ/mol) C H C-C C-N _o C-F C CI C Br 413 348 293 358...
-
Consider the simple regression model fit to the National Football League team performance data in Problem 2.1. Data From Problem 2.1 Table B. 1 gives data concerning the performance of the 26...
-
Use a financial calculator or computer software program to answer the following questions. a. What would be the future value (FV) of $19,378 invested now if the money remains deposited for eight...
-
If a firm has fixed costs of $66000 a variable cost per unit of $3 and sales price per unit of $17 what is the firm break-even point in units?
-
Which of the glycerophospholipid head groups listed in Table 9-2 can form hydrogen bonds? Table 9-2 Name of X-OH Water Ethanolamine Choline Serine myo-Inositol Glycerol Phosphatidy Iglycerol HO R -H...
-
One demonstration of P.T. Barnums maxim that theres a sucker born every minute is that new reducing aids regularly appear on the market. An eat-all-you-want nostrum, which was touted as a starch...
-
At the beginning of Year 1, the company's inventory level was stated correctly. At the end of Year 1, inventory was overstated by $2,200. At the end of Year 2, inventory was understated by $450. At...
-
a. For the allowed energies of a particle in a box to be large, should the box be very big or very small? Explain. b. Which is likely to have larger values for the allowed energies: an atom in a...
-
To have the best resolution, should an electron microscope use very fast electrons or very slow electrons? Explain.
-
X rays with a wavelength of \(0.20 \mathrm{~nm}\) undergo first-order diffraction from a crystal at a \(54^{\circ}\) angle of incidence. At what angle does first-order diffraction occur for...
-
The molecules in the rods and cones in the eye are tuned to absorb photons of particular energies. The retinal molecule, like many molecules, is a long chain. Electrons can freely move along one...
-
The spacing between atomic planes in a crystal is \(0.110 \mathrm{~nm}\). If \(12.0 \mathrm{keV} \mathrm{x}\) rays are diffracted by this crystal, what are the angles of (a) first-order and (b)...
-
If an opposing party fails to comply with discovery requests, what alternative means can be used to obtain necessary data?
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
Describe how the fumarate produced by the purine nucleotide cycle could be catabolized to CO2.
-
Calculate the ATP yield of converting the carbons of thymine to CO2.
-
Individuals with a partial deficiency of dihydropyrimidine dehydrogenase experience severe toxic effects when given high doses of the anticancer drug 5-fluorouracil. Explain.
-
Do Canadian domestic clothing manufactures who source all their inputs of labour, materials, and equipment from Canadian suppliers, have any exposure to fluctuations in exchange rates? Answer and...
-
Provide two specific examples to illustrate and explain how a Bank of Canada intervention in the foreign exchange market could stimulate growth in the Canadian Economy. Provide two specific...
-
Comparing Different Linkage Methods. The Football Bowl Subdivision (FBS) level of the National Collegiate Athletic Association (NCAA) consists of over 100 schools. Most of these schools belong to one...

Study smarter with the SolutionInn App


