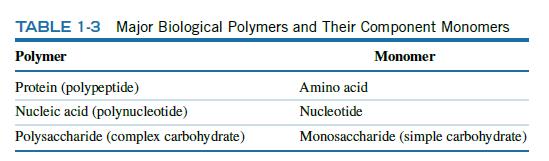
Which of the functional groups in Table 1-3 give a molecule a positive charge? Which give a
Question:
Which of the functional groups in Table 1-3 give a molecule a positive charge? Which give a molecule a negative charge?
Data from Table 1-3:
Transcribed Image Text:
TABLE 1-3 Major Biological Polymers and Their Component Monomers Polymer Monomer Protein (polypeptide) Nucleic acid (polynucleotide) Polysaccharide (complex carbohydrate) Amino acid Nucleotide Monosaccharide (simple carbohydrate)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Functional groups in Table 13 that give a molecule a positive charge Amino group NH2 Imino group NHR ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Fundamentals Of Biochemistry Life At The Molecular Level
ISBN: 9781118918401
5th Edition
Authors: Donald Voet, Judith G Voet, Charlotte W Pratt
Question Posted:
Students also viewed these Sciences questions
-
The condensation of two functional groups can result in the formation of another common functional group, which can be referred to as a compound functional group. Examine the functional groups in...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Identify all of the functional groups in each of the following compounds: (a) (b) (c) (d) (e) (f) (g) Vitamin D3 HO OMe Aspartame O NH2 NH2 Amphetamine Me Cholesterol HO OCH2CH3 Demerol CH A...
-
Photons of wavelength 1.68 cm excite transitions from the rotational ground state to the first rotational excited state in a gas. Whats the rotational inertia of the gas molecules?
-
In the Deep Creek Mining Company example described in this chapter Table, suppose again that labor is the variable input and capital is the fixed input. Specifically, assume that the firm owns a...
-
A hollow tube ABCDE constructed of monel metal is subjected to five torques acting in the directions shown in the figure. The magnitudes of the torques are T1 = 1000 lb-in., T2 = T4 = 500 lb-in., and...
-
For each of the following situations, calculate the population standard error of the mean \(\sigma \mathrm{X}^{-}\). a. \(\sigma=18 ; N=36\) b. \(\sigma=9.42 ; N=49\) c. \(\sigma=1.87 ; N=60\) d....
-
A partial list of Foothills Medical Center's costs follows: a. Cost of patient meals b. Nurses' salaries c. Depreciation of X-ray equipment d. Utility costs of the hospital e. Salary of intensive...
-
The stockholders' equity section of Company's balance sheet as of April 1 follows. On April 2, Company declares and distributes a 15% stock dividend. The stock's per share market value on April 2 is...
-
Does entropy increase or decrease in the following processes? a. b. c. d. N + 3 H 2 NH3
-
Classify the following proteins as , , or /: (a) KcsA K + channel (Fig. 10-4) Figure 10-4 (b) Thioredoxin (Fig. 23-11) Figure 23-11 Turret (a) Outer helix Selectivity filter Inner helix (b) Courtesy...
-
Write these numbers in scientific notation by counting the number of places the decimal point is moved. a. 123,456.78 b. 98,490 c. 0.000000445
-
Susan Colantuono: The Career Advice You Probably Didnt Get13:57 minutes https://www.youtube.com/watch?v=JFQLvbVJVMg How do the skills Colantuono talks about matter to measuring and implementing...
-
Let \(T_{a}^{*}=\inf \left\{t:\left|W_{t} ight|=a ight\}\). Prove that the r.v's \(T_{a}^{*}\) and \(W_{T_{a}^{*}}\) are independent and show that \(W_{T_{a}^{*}}\) is symmetric with values \(\pm a\).
-
Which National Quality Strategy priority is most important to you as a healthcare consumer, and why? Which priority do you believe is most important to providers, and why? Which priority do you...
-
Suppose you have an isolated system in which two objects about to collide have equal and opposite momenta. If the collision is totally inelastic, what can you say about the motion after the collision?
-
Prove that the projection on the \(\sigma\)-algebra \(\mathcal{F}_{g_{t}}^{+}\)of the \(\mathbf{F}\) martingale \(\left(B_{t}^{2}-t, t \geq 0 ight)\) is \(2\left(t-g_{t} ight)-t\), hence the process...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
Ex. (17): the vector field F = x i-zj + yz k is defined over the volume of the cuboid given by 0x a,0 y b, 0zc, enclosing the surface S. Evaluate the surface integral ff, F. ds?
-
Which set of binding data is likely to represent cooperative ligand binding to an oligomeric protein? (a) [Ligand] (mM) Y 0.3 0.5 0.7 0.9 (b) Ligand] (mM) 0.1 0.2 0.4 0.7 0.2 0.3 0.4 0.6 0.1 0.3 0.6...
-
Explain why a microfilament is polar whereas a filament of keratin is not.
-
Cells contain an assortment of proteins that promote microfilament disassembly during cell shape changes. How can such proteins distinguish newly synthesized microfilaments from older microfilaments?
-
Tokyo Steel's common stock currently is selling for $56 per share. the most recent dividend paid to common stockholders was $2.40, and this dividend is expected to grow at a rate of 5 percent for as...
-
Total Tools Ltd. sells 20,000 toolkits a year at an average price per set of $120. All sales are credited with terms of 2/10, net 45. The discount amount is applied to a customer's account only after...
-
The cost accountant for the Miseri Manufacturing Company has provided you with the following information for the month of June: Variable costs Per unit Total Fixed CostsDirect labor$ 2 7 . 5 0 Direct...

Study smarter with the SolutionInn App


