Consider the equation J = S E, with S as given in (4.23). If we choose
Question:
Consider the equation J = S · E, with S as given in (4.23). If we choose a Cartesian coordinate system such that
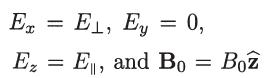
(refer to Fig. 2), verify that in this coordinate system we have
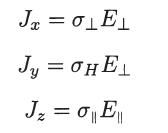
Interpret physically this result with reference to Fig. 2.
Equation.
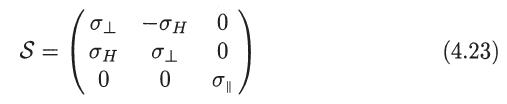
Figure 2
Transcribed Image Text:
Ex = E₁, Ey = 0, Ez = E, and Bo = Boz
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Solve the following system. 10. q = 1 (mod 3) q = 2 (mod 4) q = -5 (mod 5) q = 5 (mod 7) 0
-
Carroll, a 35 year old unmarried taxpayer with an adjusted gross income of $100,000, incurred and paid the following unreimbursed medical expenses for the year: Doctor bills resulting from a serious...
-
What are the values of m and n after executing the following code? int n = 123456789; int m= 0; while (n != 0) { } m (10 m) + (n % 10); n = n/ 10;
-
Gomez Co. had the following transactions in the last two months of its year ended December 31.Nov. 1 Paid $1,800 cash for future newspaper advertising. 1 Paid $2,460 cash for 12 months of insurance...
-
Schwenn Products, a bicycle manufacturer, uses "normal volume" as the basis for setting prices. That is, it sets prices on the basis of long-term volume predictions and then adjusts them only for...
-
What is the speed of a proton after being accelerated from \(\mathbb{N} T\) rest through a \(50 \times 10^{6} \mathrm{~V}\) potential difference?
-
During the current month, Tomlin Company incurs the following manufacturing costs: (a) Purchased raw materials of $16,000 on account. (b) Incurred factory labor of $40,000. Of that amount, $31,000...
-
Perform the following hypostasis tests. May use calculator to check answers. 1. Weyerhauser claims the average age of the trees they use to make wood and paper products is 11.5 years. It is known...
-
Assume that the average velocities of the electrons and ions in a completely ionized plasma, in the presence of constant and uniform electric (E) and magnetic (B 0 ) fields, satisfy, respectively,...
-
The April transactions of Wiset Company are described in Problem 7-2A. In Problem 7-2 Wiset Company completes these transactions during April of the current year (the terms of all its credit sales...
-
Find the area of the surface generated when the given curve is revolved about the x-axis. = = (er + e-2r) on [-2,2]
-
Consider the following pre-merger information about firm X and firm Y: Assume that firm X acquires firm Y by paying cash for all the shares outstanding at a merger premium of 5 per share. Assuming...
-
Joseph-Benoit Suvee has just been named the new chief executive officer of BluBell Fitness NV. In addition to an annual salary of 400,000, his 3- year contract states that his compensation will...
-
Shareholder Activism Many financial institutions use the media to drive shareholder activism strategies so that pressure is maximized on the target firm managers. Is this evidence that institutional...
-
You place an order for 200 units of inventory at a unit price of 95. The supplier offers terms of 2/10, net 30. (a) How long do you have to pay before the account is overdue? If you take the full...
-
You are in discussions to purchase an option on an office building with a strike price of 47 million. The building is currently valued at 45 million. The option will allow you to purchase the...
-
The high melting points of ionic solids indicate that a lot of energy must be supplied to separate the ions from one another. How is it possible that the ions can separate from one another when...
-
The unadjusted trial balance of Secretarial Services is as follows: SECRETARIAL SERVICES Unadjusted Trial Balance as at 31 December 2017 Account Debit Credit Cash at bank Office supplies Prepaid...
-
Determine the electron configuration for each of the following ions: a. A carbon atom with a negative charge b. A carbon atom with a positive charge c. A nitrogen atom with a positive charge d. An...
-
Identify which of the following two reactions you would expect to occur more rapidly: (1) addition of HBr to 2-methyl-2- pentene or (2) addition of HBr to 4-methyl-1-pentene. Explain your choice.
-
With current spectroscopic techniques (discussed in Chapters 1517), chemists are generally able to determine the structure of an unknown organic compound in just one day. These techniques have only...
-
Let's consider the Loftus Ranches investment decision. Land is $250,000. The brewery building is $1,250,000. The brewing equipment is $1,250,000. Other general start up expenses total $1,000,000. If...
-
Given the following information, determine the cost of goods sold for the period. Date Jan 1 Beginning Inventory $950 Jan 1-30 Purchases $1800 Jan 31 Ending Inventory $250.
-
Sells 11,000 umbrellas per year, selling 82,000 at 7.00 each. Fresh has approached about buying 13,000 umbrellas for 6.00 each. Would save .70 per umbrella. compared to normal production cost....

Study smarter with the SolutionInn App


