Integrate (3.49) and (3.50) to determine the particle trajectory in the plane normal to Band sketch the
Question:
Integrate (3.49) and (3.50) to determine the particle trajectory in the plane normal to Band sketch the path of the particle for q > 0 and q
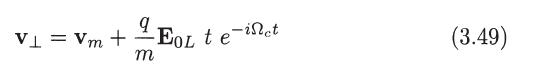
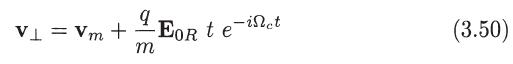
Transcribed Image Text:
V₁ = Vm + ¹/E₁L t e-inet m (3.49)
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The motion of a charged particle in an electromagnetic field can be obtained from the Lorentz equation for the force on a particle in such a field, if the electric field vector is E and the magnetic...
-
(a) Assume that f(t), in problem 4.9, is given by exp (t). Show that, in this case, (t) satisfies the Bessel equation of zero order, where = ( c /2) exp (t). Determine the two solutions of this...
-
A bubble chamber is a device that makes the path of a charged particle visible. When a charged particle with sufficient kinetic energy passes through a liquid, it will knock electrons off the atoms...
-
Exercises 11-16: For the measured quantity, state the set of numbers that most appropriately describes it. Choose from the natural numbers, integers, and rational numbers. Explain your answer....
-
Selected year- end financial statements of Cabot Corporation follow. (All sales were on credit; selected balance sheet amounts at December 31, 2012, were inventory, $ 48,900; total assets, $ 189,400;...
-
Apply the VRIO framework to determine whether Best Buy has a competitive advantage. If so, is its competitive advantage sustainable? Why or why not?
-
Rank the following from the smallest to the largest level of organization: organ, organ system, tissue, cell.
-
Clothing Frontiers began operations on January 1 and engages in the following transactions during the year related to stockholders equity. January 1 Issues 700 shares of common stock for $50 per...
-
Sales price per unit: $450 Variable cost per unit: $210 Average production: 1,000 units per month Total fixed costs: $187,000 per month How many units must Key sell in order to make a profit of...
-
Consider an electron acted upon by a constant and uniform magnetic field B = B 0 z, and a uniform but time-varying electric field E = yE y0 sin(t). Assume that the initial conditions are such that...
-
Consider the motion of an electron in the presence of a uniform magnetostatic field B = B 0 z, and an electric field that oscillates in time at the electron cyclotron frequency c , according to (a)...
-
Solve for y in terms of x. log 4 y = log 4 x log 4 10 + log 4 6
-
Darian Basemore owned an interest in five businesses in 2019. His level of participation and percentage of ownership in each enterprise is as follows: In which activity, if any, will Darian be...
-
If fixed costs are $10,000, variable costs are $4 per unit, and the target selling price per unit is $8, what is the breakeven point? (a) 2 (b) 500 (c) 2,500 (d) 4,800
-
Sandy had the following items on her timely filed 2018 income tax return: Sandy inadvertently omitted some income on her 2018 return. What is the statute of limitations if she omitted $100,000 of...
-
The following random sample is taken from a normal population. (a) If the population man is = 60, what is t statistics for the sample? (b) If the population man is = 55, what is t statistics for...
-
Brent Fullback owned four passive activity interests in 2019: On March 2, 2019, Fullback sold his entire interest in A-1 for $15,000. His basis in the activity on January 1, 2019, was $11,000....
-
The heat required to raise the temperature from 300.0 K to 400.0 K for 1 mole of a gas at constant volume is 2079 J. The internal energy required to heat the same gas at constant pressure from 550.0...
-
Express mass density in kg/m3 and weight density in lb/ft3. 1. Find the mass density of a chunk of rock of mass 215 g that displaces a volume of 75.0 cm3 of water. 2. A block of wood is 55.9 in. x...
-
Compound A has molecular formula C 5 H 10 . Hydroboration-oxidation of compound A produces a pair of enantiomers, compounds B and C. When treated with HBr, compound A is converted into compound D,...
-
Consider each pair of compounds below, and determine whether the pair represent the same compound, constitutional isomers, or different compounds that are not isomeric at all: a. b. c. d.
-
What reaction will take place if H 2 O is added to a mixture of NaNH 2 /NH 3 ?
-
The artificial intelligence value chain may be interpreted differently for the same industry. Please think through how this would apply to the following: You are the Director of Information...
-
Make two diagrams that describe in detail: The importance of considering various job analysis strategies. How can this strategy impact the results and performance of the organization? What would be...
-
Project supply chain performance can be improved by careful and innovative use of partnering, third-party involvement, lean purchasing, sourcing, logistics, and information. Elucidate how lean...

Study smarter with the SolutionInn App


