Question: Complete this table for H 2 O: T, C 140 125 300 P, kPa 550 750 V, m/kg 0.035 0.140 Phase description Saturated liquid
Complete this table for H2O:
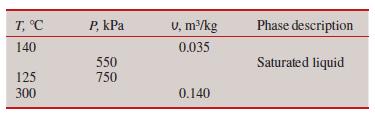
T, C 140 125 300 P, kPa 550 750 V, m/kg 0.035 0.140 Phase description Saturated liquid
Step by Step Solution
3.51 Rating (164 Votes )
There are 3 Steps involved in it
Complete the following table for H 2 O EES SOLUTION Given ... View full answer

Get step-by-step solutions from verified subject matter experts


