A 1-m 3 rigid tank has propane at 100 kPa, 300 K and connected by a valve
Question:
A 1-m3 rigid tank has propane at 100 kPa, 300 K and connected by a valve to another tank of 0.5 m3 with propane at 250 kPa, 400 K. The valve is opened, and the two tanks come to a uniform state at 325 K. What is the final pressure?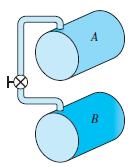
Transcribed Image Text:
A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
We shall assume that propane behaves as an ideal gas ie it satisfies the ideal gas equation P Vn R T ...View the full answer

Answered By

Tarang Hirapra
I have done my bachelors in Mechanical Engineering. After that I home-schooled high school students for about a year. I also have experience of about 6 months in freelancing for tutoring in online education platforms.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A 1 m3 rigid tank has propane at 100 kPa, 300 K and connected by a valve to another tank of 0.5 m3 with propane at 250 kPa, 400 K. The valve is opened and the two tanks come to a uniform state at 325...
-
A 1 m3 rigid tank has propane at 100 kPa, 300 K and connected by a valve to another tank of 0.5 m3 with propane at 250 kPa, 400 K. The valve is opened and the two tanks come to a uniform state at 325...
-
A 35 ft3 rigid tank has propane at 15 psia, 540 R and connected by a valve to another tank of 20 ft3 with propane at 40 psia, 720 R. The valve is opened and the two tanks come to a uniform state at...
-
Find dy/dx for the following functions. y = sin x + cos x
-
Braxton Sebastian manages a computer software distributorship and offers a desktop publishing software package for $395 with a trade discount series of 5/5/8. What is the trade discount on this...
-
A firm is set up to produce and sell cotton shirts. It buys plant and machinery for $2 million and land for $1 million, and constructs a warehouse for another $1 million. Each year, it hires 100...
-
The employees of the Telektronics Corporation have six-digit identification numbers that are randomly generated. Determine whether the data described are qualitative or quantitative and give their...
-
Peterborough Medical Devices makes devices and equipment that it sells to hospitals. The organization has a profit-sharing plan that is worded as follows: The company will make available a...
-
ok t nces Pizza Express Incorporated began the Year 2 accounting period with $7,500 cash; $5,000 of common stock; and $2,500 of retained earnings. Pizza Express was affected by the following...
-
The following data report total, monthly U.S. book-store sales in millions of dollars fromJanuary 2016 to March 2019. (Go to https://www.census.gov/retail/index.html#mrts, find Monthly Retail Trade...
-
Air in an internal-combustion engine has 227C, 1000 kPa, with a volume of 0.1 m 3 . Combustion heats it to 1800 K in a constant-volume process. What is the mass of air, and how high does the pressure...
-
Ammoniain a piston/cylinder arrangement is at 700 kPa and 80C. It is now cooled at constant pressure to saturated vapor (state 2), at which point the piston is locked with a pin. The cooling...
-
What is the current status of the Sarbanes-Oxley Act? What do executives think of it now? What impact has it had?
-
Explain the education at the factory According to this video- https://www.youtube.com/watch?v=xMm-YMO5H7o
-
Translate the given loop to assembly language: int p = 0, m = 500; for (int i = 1; i < 10; i++) { } m += (p/2) p+6; cout < < l; for (int j = 0; j <5; j++) { } cout < < (p %10);
-
Perform the indicated operations and simplify the complex fraction one of the following factoring formulas: 2-y = (x - y) (x + xy + y) r+y = (x+y)(x - xy + y) -((h) (a(2) + a * h)) h completely. You...
-
From the following selected balances and additional information, prepare the adjusting entries at the end of the fiscal year May 31: Debit Credit Accounts Receivable 18% Notes Receivable Supplies...
-
1. What constitutes "ethical management" in ethical nonprofit organizations? What are the five core concepts that define it? please explain all five core concepts. 2 What are the key characteristics...
-
Answer the following questions: a. What is the relationship between chemical shift in ppm and operating frequency? b. What is the relationship between chemical shift in hertz and operating frequency?...
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
A hydraulic cylinder of area 0.01 m2 must push a 1000 kg arm and shovel 0.5 m straight up. What pressure is needed and how much work is done?
-
Consider a household refrigerator that has just been filled up with room-temperature food. Define a control volume (mass) and examine its boundaries for rates of work and heat transfer, including...
-
A room is heated with an electric space heater on a winter day. Examine the following control volumes, regarding heat transfer and work , including sign. a) The space heater. b) Room c) The space...
-
JR's Cleaning Solutions provides Sally's Janitorial Services all the cleaning products they require on a monthly basis. How would you describe JR's Cleaning Solutions and Sally's relationship?
-
Determine the total and unit target cost to manufacture, sell, distribute, and service the portable brushes. Begin by computing the total target cost to manufacture, sell, distribute, and service the...
-
Calculate the NPV of the Project using a discount rate of 15%. Year 0 = -$100; Year 1 = $50; Year 2 = $40; Year 3 = $40; Year 4= $15.

Study smarter with the SolutionInn App


