A tank has two sides initially separated by a diaphragm. Side A contains 1 kg water and
Question:
A tank has two sides initially separated by a diaphragm. Side A contains 1 kg water and side B contains 1.2 kg air, both at 20◦C, 100 kPa. The diaphragm is now broken and the whole tank is heated to 600◦C by a 700◦C reservoir. Find the final total pressure, heat transfer, and total entropy generation.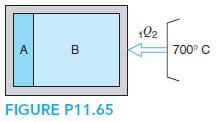
Transcribed Image Text:
102 A. B 700° C FIGURE P11.65
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
Final total pressure 83549 kPa Heat transfer 822 kJ Total entropy generation 578 JK The final total ...View the full answer

Answered By

Firoz K
I have extensive experience in education and tutoring, having worked as a tutor for the past three years in both group and individual settings. During my time as a tutor, I have successfully helped students improve their academic performance in a variety of subjects, including mathematics, science, language arts, and social studies. I have also developed and implemented personalized learning plans and differentiated instruction techniques to accommodate the individual needs of my students. Moreover, I have effectively communicated with parents and teachers to ensure that the students receive the best possible education and guidance. My strong organizational, communication, and problem-solving skills have enabled me to successfully collaborate with students, parents, and teachers in order to provide an effective and enjoyable learning experience.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A piston/cylinder contains 1 kg water at 20oC with volume 0.1 m3. By mistake someone locks the piston preventing it from moving while we heat the water to saturated vapor. Find the final temperature,...
-
A piston cylinder contains 1 kg water at 20oC with a constant load on the piston such that the pressure is 250 kPa. A nozzle in a line to the cylinder is opened to enable a flow to the outside...
-
A piston/cylinder contains 1 kg water at 150 kPa, 20C. The piston is loaded so pressure is linear in volume. Heat is added from a 600C source until the water is at 1 MPa,...
-
What would a profile look like across a restraining bend? Releasing bend?
-
Apply the rank order clustering technique to the part-machine incidence matrix in the following table to identify logical part families and machine groups. Parts are identified by letters, and...
-
Calculate the number of cations, anions, and formula units in a unit cell of each of the following solids: (a) The cesiumchloride unit cell shown in Fig. 3H.30; (b) The rutile (TiO 2 ) unit cell...
-
Provide an example of an economic objective of the tax law.
-
Slug flow is an idealized tube flow condition for which the velocity is assumed to be uniform over the entire tube cross section. For the case of laminar slug flow with a uniform surface heat flux,...
-
The following balance sheet for the Los Gatos Corporation was prepared by a recently hired accountant. In reviewing the statement you notice several errors. LOS GATOS CORPORATION Balance Sheet At...
-
Draw the free-body diagram for the following problems. a) The beam in Prob. 515. b) The linkage in Prob. 516. c) The frame in Prob. 517. d) The beam in Prob. 518. -8 m- 75 N/m 800 Nm
-
If I have air at 100 kPa and (a) 10C, (b) 45C, (c) 110C, what is the maximum absolute humidity I can have?
-
A mixture of 60% helium and 40% nitrogen by mass enters a turbine at 1 MPa, 800 K at a rate of 2 kg/s. The adiabatic turbine has an exit pressure of 100 kPa and an isentropic efficiency of 85%. Find...
-
Suppose that the Dallas School District wants to achieve Six Sigma quality levels of performance in delivering students to school. It has established a 20-minute window as an acceptable range within...
-
evaluate the relationship of the post's content compared to module concepts and materials, and other relevant sources deemed meaningful. The overall goal for your discussion engagement is to push...
-
A. Introduction: The introductory part must contain the following. 1. Concept of knowledge management. (Provide minimum 3 definitions of knowledge management by well-known authors) followed by...
-
What are the theoretical models, concepts, arguments, and ideas that have been developed in relation to consumers who prefer to consume Puerto Rico's rich rice brand, rather than other brands...
-
Lockheed Martin is sourcing aircraft parts from Boeing after exploring other defense partners in the industry. This process is typically high risk due to sensitivity of the information being...
-
Explain the importance of organizational behavior in the effective administration of criminal justice organizations.
-
Explain the following facts: a. 1-Hexanol has a higher boiling point than 3-hexanol. b. Diethyl ether has very limited solubility in water, but tetrahydrofuran is essentially completely soluble....
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Below are NMR spectra of several compounds. Identify whether these compounds are likely to contain ethyl, isopropyl, and/or tert-butyl groups: (a) (b) (c) (d) Proton NMR CH12 5 Chemical Shift (ppm) 6
-
Below are NMR spectra of several compounds. Identify whether these compounds are likely to contain ethyl, isopropyl, and/or tert-butyl groups: (a) (b) (c) (d) Proton NMR C3H12 Chemical Shift (ppm)...
-
Each of the following compounds exhibits a 1 H NMR spectrum with only one signal. Deduce the structure of each compound: (a) C 5 H 10 (b) C 5 H 8 C l4 (c) C 12 H 18
-
A 19-year bond with a 11.8 percent semiannual coupon and a $1,000 face value has a nominal yield to maturity of 9.9 percent. The bond currently sells for $1,161.31. The bond, which may be called...
-
Suppose you are the financial manager of a firm considering the following five projects (expand below to see the five projects). Five Projects Under Construction Five Projects Under Consideration...
-
We sign a contract that calls for a down payment of 3 , 0 0 0 and for payments of 2 5 0 at the end of each month for 6 years. Money is worth 1 2 % compounded monthly. If we miss the first 8...

Study smarter with the SolutionInn App


