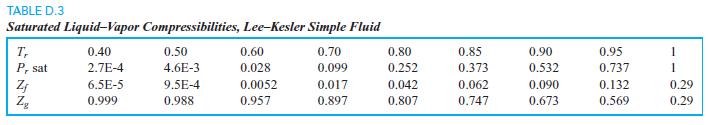
For Tr = 0.7, what is the ratio vg/vf using Fig. D.1 compared to Table D.3? TABLE
Question:
For Tr = 0.7, what is the ratio vg/vf using Fig. D.1 compared to Table D.3?
Transcribed Image Text:
TABLE D.3 Saturated Liquid-Vapor Compressibilities, Lee-Kesler Simple Fluid 0.40 0.70 T, P, sat 0.50 0.60 0.80 0.85 0.90 0.95 1 2.7E-4 4.6E-3 0.028 0.099 0.252 0.373 0.532 0.737 1 6.5E-5 9.5E-4 0.0052 0.017 0.042 0.062 0.090 0.132 0.29 0.999 0.988 0.957 0.897 0.807 0.747 0.673 0.569 0.29
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
In Fig D1 we can see that for Tr 07 the ratio of vgvf is appro...View the full answer

Answered By

MERCY NGEI
Online education is a form of education that is delivered through the internet. Online education can take many forms, including webinars, online courses, and virtual classrooms. Continuing education includes any type of learning that takes place after formal education, such as professional development courses, workshops, and training programs. Higher education includes college, university, and vocational training programs. Higher education can lead to degrees and certifications in a wide range of fields, including science, technology, engineering, arts, and humanities.: Secondary education typically includes grades six or seven through twelve, depending on the country. Secondary education focuses on a more advanced curriculum that prepares students for higher education or vocational training.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
What is the ratio of rates of effusion of N2 and O2 under the same conditions?
-
provide formulas, solutions, and explanations in full detail An acid with a pKa of 8.0 is present in a solution with a pH of 6.0. What is the ratio of the protonated to the deprotonated form of the...
-
What is the ratio of protons to electrons in an atom, and what is the net electric charge of an atom?
-
In a two-player, one-shot simultaneous-move game each player can choose strategy A or strategy B. If both players choose strategy A, each earns a payoff of $500. If both players choose strategy B,...
-
Is there a significant difference in the level of community service participation between college and high school graduates? According to the 2010 GSS, 38% of 160 college graduates reported...
-
Explain why the density of vanadium (6.11 g cm 3 ) is significantly less than that of chromium (7.19 g cm 3 ). Both vanadium and chromium crystallize in a body-centered cubic lattice.
-
Let \(A\) be an increasing process, with \(\mathbb{Q}\) and \(\mathbb{P}\) as above. Prove that the \(\mathbb{Q}\)-compensator of \(\int_{0}^{t} \frac{d A_{s}}{L_{s}}\) is \(\int_{0}^{t} \frac{d...
-
A thermocouple has the following characteristics when it is immersed in a stirred bath: Mass of thermocouple 1g. Heat capacity of thermocouple = 0.25 cal/g C I-feat transfer coefficient = 20 cat/cm2...
-
2. In January 2016, the Free Cancer Foundation accepted an endowment of $500,000, the income from which is restricted to promoting research related to recovery from cancer. All gains, whether...
-
Baseballs vary somewhat in their rebounding coefficient. A baseball that has a large rebound coefficient will travel farther when the same force is applied to it than a ball with a smaller...
-
To solve for v given (P, T) in Eq. 2.14, what is the mathematical problem? RT P = - v - b a (2.14) v? + cbv + db
-
Find the constants in the curve fit for the saturation pressure using Wagners correlation, as shown in the previous problem for water and methane. Find other correlations in the literature, compare...
-
In Problems 1724, express each sum or difference as a product of sines and/or cosines. sin (40) + sin(20)
-
Suppose the return on the market portfolio is 12.80% and the risk-free rate is 5.30%. If the standard deviation of the market portfolio is 16.00%, what is the standard deviation of a stock with an...
-
5. Work through this hypothesis test using each step of hypothesis testing. You may want to borrow the promptsfrom the previousproblem A sampleof 77 homes for salein ABC City showeda meanaskingprice...
-
evaluates the effectiveness of current levels of organisational corporate communications for a named organisation.
-
A company's non-callable bonds currently sell for $1,165. They have a 15-year maturity, a coupon rate of 8% with semiannual payments, and a par value of $1,000. What is their yield to maturity ? How...
-
According to Stewart (2003), what is "wrong" with accounting, and how did this contribute to the corporate scandals of the early 2000s? What recommendations does he make for "fixing" the problem?
-
Telescopes used in astronomy have large lenses (or mirrors). One reason is to let a lot of light in important for seeing faint astronomical bodies. Can you think of another reason why it is an...
-
Troy is a qualified radiologist who operates a successful radiology practice from purpose- built rooms attached to his house. Troy works in the practice three days a week, and the other two days he...
-
2.25 moles of an ideal gas with C V ,m = 3/2 R undergoes the transformations described in the following list from an initial state described by T = 310.K. and P = 1.00 bar. Calculate q, w, U, H, and...
-
Compounds A, B, C, and D are constitutionally isomeric, aromatic compounds with molecular formula C 8 H 10 . Deduce the structure of compound D using the following clues. The 1 H NMR spectrum of...
-
Consider the reversible Carnot cycle shown in Figure 5.2 with 1.25 mol of an ideal gas with C V = 5/2R as the working substance. The initial isothermal expansion occurs at the hot reservoir...
-
Use the Limit Comparison Test to determine the convergence or divergence of the series. lim n 00 n = 1 6n+1 9 6 +1 = L > 0
-
Write the following expression as a single summation in terms of k. k = 1 k k + 2 ? m + 1 + == m+3 k = 1
-
Sally needs to record a sales tax deposit of $375.23. What steps should she take to record this deposit? How they can be recorded in QBO.

Study smarter with the SolutionInn App


