Show that the van der Waals equation can be written as a cubic equation in the compressibility
Question:
Show that the van der Waals equation can be written as a cubic equation in the compressibility factor, as in Eq. 12.53.
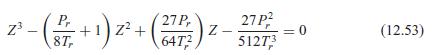
Transcribed Image Text:
2 () () - 27P, +1) z+ 6472 27P? 51273 z - = 0 (12.53)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The van der Waals equation of state is given by P aV2 V b RT where P is the pressure V is the molar ...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Show that van der Waals equation can be written as a cubic equation in the compressibility factor involving the reduced pressure and reduced temperature as (27 P 27 P2 512 T, (T+1) z2- - 1) z - Z3 ST...
-
Show that the van der Waals equation of state does not satisfy the criteria of intrinsic stability for all values of the parameters. Sketch the curves of P versus V for constant T (the isotherms of...
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
Parent Ltd owns 80% of Subsidiary Ltd. In the financial year ended 30 June 20X2, Subsidiary Ltd sold inventory to Parent Ltd. Details regarding the transaction are as follows: Cost to Subsidiary to...
-
What type of exchange rate system was the gold standard? Explain how it operated?
-
Formamide, HC(O)NH 2 , is prepared at high pressures from carbon monoxide and ammonia, and serves as an industrial solvent (the parentheses around the O indicate that it is bonded only to the carbon...
-
Why are some archaea described as extremophiles?
-
On January 1, 2014, Mixon Co. borrowed cash from First City Bank by issuing a $90,000 face value, three-year term note that had a 7 percent annual interest rate. The note is to be repaid by making...
-
From How to Invest in Stocks: A Beginner's Guide: The article notes when choosing a brokerage you should consider what ?
-
Harris Company must set its investment and dividend policies for the coming year. It has three independent projects from which to choose, each of which requires a $3 million investment. These...
-
Show how to find the constants in Eq. 12.52 for the van der Waals EOS. Vc = 3b 27 R?T? a 64 P. (12.52) RT. b 8P.
-
Find the heat of evaporation, hfg, for R-134a at 0C from the generalized charts and compare to the value in Table B.5.
-
The Runge Controls Corporation manufactures and markets electrical control systems: temperature controls, machine controls, burglar alarms, and the like. The company acquires electrical and...
-
Are there certain types of IG Reels that tend to perform better than others? What is the insights regarding the use of hashtags?
-
In the class, they hold opposing views on an emotionally charged political or social issue, for example, private medical clinics, mandatory retirement, legalization of marijuana, or patients' right...
-
Stock A beta = 1,25 and Stock B beta = 0.75. CAPM applies. Expected Return on market portfolio = 10% and Risk free rate of return = 3% 1. What is the expected return of the portfolio consisting of...
-
Amazon has reinvented itself again and again and thrived on organizational change. Since the company went online in 1995, the e-commerce giant has evolved under the leadership of one personJeff...
-
Consider a certain type of machinery that you can buy in the USA for $60,000 and in Japan for 6,780,000 yen. The current exchange rate is 0.01 dollar per yen. Carefully following all numeric...
-
Suppose some astronauts have landed on Mars. (a) When Mars and Earth are on the same side of the Sun and as close as they can be to one another, how long does it take for radio transmissions to...
-
San Carlos Bank and Trust Company uses a credit-scoring system to evaluate most consumer loans that amount to more than $2,500. The key factors used in its scoring system are found at the conclusion...
-
For a pair of keto-enol tautomers, explain how IR spectroscopy might be used to identify whether the equilibrium favors the ketone or the enol.
-
Acrolein is an α,β-unsaturated aldehyde that is used in the production of a variety of polymers. Acrolein can be prepared by treating glycerol with an acid catalyst. Propose...
-
Draw the structure of the product with molecular formula C 10 H 10 O that is obtained when the compound below is heated with aqueous acid. CN CN C10H100 Heat
-
Tesla's recent balance sheet to describe the different forms of liabilities it uses, make sure to break down Tesla's company's debt. Is the debt increasing or decreasing? Does Tesla have enough...
-
When a QuickBooks Desktop Enterprise 20.0 or newer user adds inventory parts to a purchase order and then clicks the View Vendor Price button on the main ribbon of the displayed purchase order, what...
-
It is important for a public health professional to show evidence of professional development over time. A professional portfolio is one way to collect and showcase achievements and artifacts or...

Study smarter with the SolutionInn App


