The cogenerating power plant shown in Problem 9.80 burns 170 kg/s air with natural gas, CH 4
Question:
The cogenerating power plant shown in Problem 9.80 burns 170 kg/s air with natural gas, CH4. The setup is shown in Fig. P13.205, where a fraction of the air flow out of the compressor with pressure ratio 15.8:1 is used to preheat the feedwater in the steam cycle. The fuel flowrate is 3.2 kg/s. Analyze the system, determining the total heat transfer to the steam cycle from the turbine exhaust gases, the heat transfer in the preheater, and the gas turbine inlet temperature.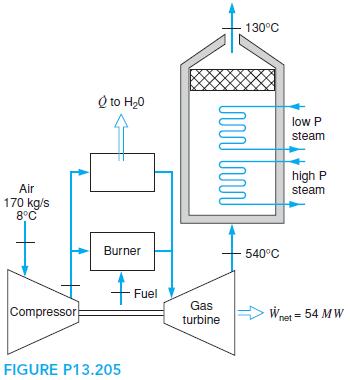
Transcribed Image Text:
130°C Q to H20 low P steam high P Air steam 170 kg/s 8°C Burner 540°C Fuel Gas Compressor Wnet = 54 MW turbine FIGURE P13.205
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
In order to analyze the system and determine the total heat transfer to the steam cycle from the turbine exhaust gases the heat transfer in the prehea...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
The simple steam power plant shown in Problem 6.39 has a turbine with given inlet and exit states. Find the availability at the turbine exit, state 6. Find the second law efficiency for the turbine,...
-
The simple steam power plant shown in Problem 6.99 has a turbine with given inlet and exit states. Find the availability at the turbine exit, state 6. Find the second law efficiency for the turbine,...
-
The asterisk in the structure shown in Problem 9 indicates the site where the base is attached via a two-carbon linker. Compare the overall backbone length for a PNA and a DNA molecule and explain...
-
As mentioned in Section 5.6, Sainte-Venants principle will allow particular boundary conditions to be replaced by their statically equivalent resultant. For problems (b), (c), (d),and (f) in Exercise...
-
Solve previous Problem 25.12 except that the current inventory on hand and on order for S3, C6, and M6 is as follows: for S3, inventory on hand is 2 units and quantity on order is zero; for C6,...
-
Find (a) The network function H(s) = V 2 /V 1 in the circuit shown in Fig. 12-13, (b) H(j) for LC = 2/ 0 2 and L/C = R 2 , and (c) The magnitude and phase angle of H( j) in (b) For 0 = 1 rad/s. + V...
-
Three identical resistors have an equivalent resistance of \(30 \Omega\) when connected in parallel. What is their equivalent resistance when connected in series?
-
Karen Corporation has compiled the following information from the accounting system for the one product it sells: Sales price . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $900...
-
Describe a new content analysis or engagement tool that you learned about. What are its main benefits? How could this tool be used in relation to HP's marketing analytics listening program?
-
Analyzing, Forecasting, and Interpreting Both Income Statement and Balance Sheet Following are the income statements and balance sheets of Best Buy Co., Inc. Income Statement, Feb. 26, 2011 Feb. 27,...
-
Study the coal gasification process that will produce methane, CH 4 , or methanol, CH 3 OH. What is involved in such a process? Compare the heating values of the gas products with those of the...
-
A car that runs on natural gas has it stored in a heavy tank with a maximum pressure of 3600 psi (25 MPa). Size the tank for a range of 300 miles (500 km), assuming a car engine that has a 30%...
-
In this exercise, you will modify the program from Lab 9-2 in Chapter 9. If necessary, create a new project named ModifyThis13 Project, and save it in the Cpp8\Chap10 folder. Copy the instructions...
-
1. In your own words, summarize the origins of law enforcement in the Canada (minimum 500 words). 2. Select one police agency operating in the area where you live. Describe its organizational...
-
PART 1: ECONOMIC PLAN 1. What are two economic goals? Choose only most important ones, recognizing trade offs. 2. Choose two products/services to be produced? a. How will they be produced? 98 b. For...
-
the Coca-Cola Company Each associate brings his or her unique talents and ideas to work every day to help the CocaCola system achieve the goals outlined in our 2020 Vision. Associates also represent...
-
How is your Costco mission statement associated with its competitive advantage? Does Nike seem to have any longer-term challenging or stretch goals that would serve as its strategic intent? Is...
-
The distances between the study stations are presently gotten to the necessary precision. Steel tapes might be laid along the ground to gauge the inclination lengths, while vertical points might be...
-
Indicate whether each of the following structures is (R)-2-chlorobutane or (S)-2 chlorobutane. (Use models, if necessary.) a. b. c. d. e. f. Cl HHCH H3C CHi Cl CH3 CH3 CH CH3 CHs CI Cl
-
What are the four types of poultry production systems? Explain each type.
-
Liquid n-butane at T0, is sprayed into a gas turbine with primary air flowing at 1.0 MPa, 400 K in a stoichiometric ratio. After complete combustion, the products are at the adiabatic flame...
-
Butane gas at 25C is mixed with 150% theoretical air at 600 K and is burned in an adiabatic steady flow combustor. What is the temperature of the products exiting the combustor?
-
Natural gas, we assume methane, is burned with 200% theoretical air and the reactants are supplied as gases at the reference temperature and pressure. The products are flowing through a heat...
-
On a qualitative basis, plot minor species concentrations (OH, H, and O) versus time for hydrogen-oxygen mixture kept in a constant volume vessel for case (a) mixture does not explore, and (b)...
-
We all have our own biases that we may not even realize we have our implicit biases. Harvard's Project Implicit website provides a test that you can use to explore the implicit biases you may have....
-
Air expands through a turbine from 7bar, 960 K to 1.0 bar, 450 K. The inlet velocity is small compared to the exit velocity of 75 m/s. The turbine operates at steady state and develops a power output...

Study smarter with the SolutionInn App


