C 6 H 4 NH 2 COOH, para-aminobenzoic acid (PABA), is used in some sunscreen agents. Calculate
Question:
C6H4NH2COOH, para-aminobenzoic acid (PABA), is used in some sunscreen agents. Calculate the concentrations of hydronium ion and para-aminobenzoate ion, C6H4NH2COO-, in a 0.080 M solution of the acid. The value of K a is 2.2 × 10-5.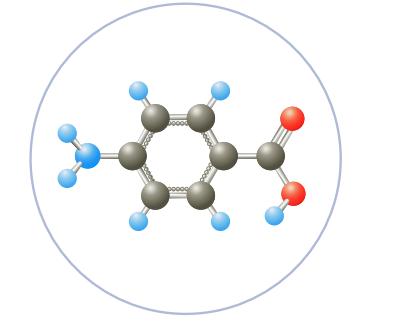
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
To solve assemble a table of starting change and equilibrium concen...View the full answer

Answered By

Ajeet Singh
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
4+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
C6H4NH2COOH, para-aminobenzoic acid (PABA), is used in some sunscreen agents. Calculate the concentrations of hydronium ion and para-aminobenzoate ion, C6H4NH2COO, in a 0.055 M solution of the acid....
-
A solution of barium hydroxide at 25C is 0.125 M Ba(OH)2. What are the concentrations of hydronium ion and hydroxide ion in the solution?
-
Barbituric acid, HC4H3N2O3, is used to prepare various barbiturate drugs (used as sedatives). Calculate the concentrations of hydronium ion and barbiturate ion in a 0.10 M solution of the acid. The...
-
Show that if powers of x greater than x 5 are neglected. In sin x X =-=-x-x 180
-
Refer to the International Conference on Web Intelligence and Intelligent Agent Technology (2010) study of how social media (e.g., Twitter.com) may influence the products consumers buy, Recall that...
-
For the holiday season of 2017, nearly 59 percent of consumers planned to buy gift cards. According to the National Retail Federation, millennials like to purchase gift cards (Dayton Daily News...
-
Consider the flowsheet for the manufacture of vinyl chloride in Figure 2.6. (a) If the pyrolysis furnace and distillation towers are operated at low pressure \((1.5 \mathrm{~atm})\), what are the...
-
Rosario Department Store uses a perpetual inventory system. Data for product E2-D2 include the following purchases. On June 1, Rosario sold 26 units, and on August 27, 40 more units. Prepare the...
-
Q1. Why wheel bearing lubrication is important? Q2. Explain the steps involved in wheel bearing adjustment procedure? Q3. What are the post fitting procedures and checks of wheel hubs and associated...
-
(bor a fixed system that is independent of time &, show that the kinetic energy Tis a homogeneous quadratic function of the generalized veiocities dj nad obeys the Euler's Theorem forin = 2.
-
Formic acid, HCHO 2 , is used to make methyl formate (a fumigant for dried fruit) and ethyl formate (an artificial rum flavor). What is the pH of a 0.12 M solution of formic acid? What is the degree...
-
A solution of acetic acid, HC 2 H 3 O 2 , on a laboratory shelf was of undetermined concentration. If the pH of the solution was found to be 2.57, what was the concentration of the acetic acid? The K...
-
Progressive Parts Corporation issued 25,000 preferred shares on May 10 for $35 each. Each share is convertible into two common shares. On November 21, the preferred shares had a fair value of $37...
-
3. (10) The script below demonstrates a very basic loop structure. Indicate the value of R1 after 5 iterations of the loop. (Given the very first line of code, you can assume R1 starts out as 0x00)...
-
An airplane flies 450 km/h at an elevation of 2000 m in a standard atmosphere. (a) Detemrine the pressure at point 1 that is far ahead of the airplane. (b) Determine the pressure at the stagnation...
-
You are a police officer examining the scene of a car crash that claimed the life of someone driving a Tesla. There was a collision involving them and a pickup truck. The truck driver, who survived,...
-
The Art of Not Being Governed" (18 min. 9 sec.) by Yale University Located at: https://youtu.be/aVwrUsib4vU?si=wpPMAgu6HKV_RuEA 1. Who are the 'hill people,' and why are they not 'primitive'? 2. How...
-
Ann and Tom want to establish a fund for their grandson's college education. What lump sum must they deposit at a 6% annual interest rate, compounded quarterly, in order to have $60,000 in the fund...
-
(a) How far does the moon in Example 28.1 travel according to the Earth-bound observer? (b) How far does it travel as viewed by an observer moving with it? Base your calculation on its velocity...
-
A. Select a recent issue (paper or online) of Report on Business Magazine, Canadian Business Magazine (online only), Bloomberg Businessweek, Fast Company, The Economist, or another business magazine....
-
The following data were collected for the reaction A(g) + B(g) Products. a. Determine the rate law for this reaction. b. Calculate the rate constant. c. Calculate the rate when [A] = 0.200 M and [B]...
-
Dinitrogen pentoxide, N2O5, undergoes first-order decomposition in chloroform solvent to yield NO2 and O2. The rate constant at 45oC is 6.2 104/min. Calculate the volume of O2 obtained from the...
-
Hydrogen peroxide undergoes a first-order decomposition to water and O2 in aqueous solution. The rate constant at 25oC is 7.40 104/s. Calculate the volume of O2 obtained from the decomposition...
-
Q5 Financial maths Initial investment: E 25,000 Year 1 return: 8,000 Year 2 return: E 15,000 Year 3 return: E Year 4 return: E 10,000 5,000 (i) Enter appropriate formulae in the shaded cells in the...
-
The production manager of Rordan Corporation has submitted the following quarterly production forecast for the upcoming fiscal year: Units to be produced 1st Quarter 10,000 2nd Quarter 7,500 3rd...
-
Annswer the following questions: [10 marks] A. Define two of the main advantages of explicit architecture. B. Compare between (1) Model-View-Controller (MVC), (1) Layered architecture pattern. The...

Study smarter with the SolutionInn App


