Using the molecular models, name the following chemical compounds: F Se B F F H H b
Question:
Using the molecular models, name the following chemical compounds:
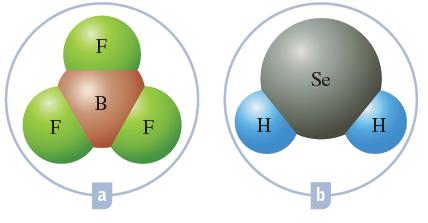
Transcribed Image Text:
F Se B F F H H b a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a Boron ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
You perform a chemical reaction using the hypothetical elements A and B. These elements are represented by their molecular models shown below: The product of the reaction represented by molecular...
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
In Problems 530, a. Classify the sequences as arithmetic, geometric, Fibonacci, or none of these. b. If arithmetic, give d; if geometric, give r; if Fibonacci, give the first two terms; and if none...
-
A rectangle has a perimeter of 38 m and an area of 84 m2. What are the dimensions of the rectangle?
-
Why do you think alcohol is the mostly widely abused drug in the United States? How can we better communicate the dangers and challenges associated with heavy and/or sustained alcohol use? According...
-
For each of the following situations, calculate the \(z\)-statistic \((z)\). a. \(\mathrm{X}^{-}=8.00 ; \mu=5 ; \sigma=6 ; N=16\) b. \(\mathrm{X}^{-}=4.00 ; \mu=2 ; \sigma=8 ; N=25\) c....
-
The records of Hollywood Company reflected the following balances in the stockholders equity accounts at December 31, 2013: Common stock , par $12 per share, 50,000 shares outstanding Preferred...
-
Assume a market price is set artificially low. In other words, the price is set below the equilibrium price. How will this affect the market
-
A ride hailing company has their DB structured in 3 major tables as described in the SCHEMA section below. Write a query to fetch the top 100 users who traveled the most distance using the service....
-
Name the following compounds: a. Cl 2 O 6 , b. PCl 3 , c. PCl 5 .
-
What are the name and formula of the anion corresponding to perbromic acid, HBrO 4 ?
-
Talkington Electronics issues a $400,000, 8%, 15-year mortgage note on December 31, 2019. The proceeds from the note are to be used in financing a new research laboratory. The terms of the note...
-
and the 5. (a) Find the tension in each string in figures (a) through compression in the beams of (c), (d)). Assume that the strings and beams are weightless. Heavy lines indicate ceilings, walls, or...
-
A pool ball is rolling along a table with a constant velocity. The components of its velocity vector are v = 0.4m/s and v = 0.6m/s. Calculate the distance it travels in 0.4 s Express your answer in...
-
An asset that cost $25,000 was retired and sold for $16,000 cash. Accumulated depreciation on the asset was $14,000. The entry to record this retirement and sale calls for recognizing: a. no gain or...
-
(a) Determine the electric field strength at a point 1.00 cm to the left of the middle charge shown in the figure below. (Enter the magnitude of the electric field only.) 6.00 C 1.50 N/C + 3.00 cm...
-
Average trade receivables of a firm are 40,000t, average finished goods are 50.000, cost of goods sold is 200.000t and net sales is 250.000t. What is trade receivables turnover?
-
Consider the large-sample level .01 test in Section 8.4 for testing H0: p = .2 against Ha: p > .2. a. For the alternative value p = .21, compute (.21) for sample sizes n = 100, 2500, 10,000, 40,000,...
-
The following table shows the rates of total return in successive years from 2004 to 2008 for the Sprott Canadian Equity Fund and for the benchmark Toronto Stock Exchange S&P/TSX Composite Index. By...
-
Potassium chromate is an important compound of chromium. It is composed of K+ and CrO42 ions. Write the formula of the compound.
-
Write the names of the following compounds: a. CaO, b. PbCrO4.
-
Like Dalton, chemists continue to model atoms using spheres. Modern models are usually drawn with a computer and use different colors to represent atoms of different elements. Which of the models...
-
How can bash shell scripting improve resource utilization and process management in Unix systems?
-
A solid sphere that is uniformly positively charged produces an electric field. Assume no other objects are around. What is the magnitude of the electric field a distance r from the center of the...
-
Why is potential difference important in x - ray production?

Study smarter with the SolutionInn App


