What hybrid orbitals would be expected for the central atom in each of the following molecules or
Question:
What hybrid orbitals would be expected for the central atom in each of the following molecules or ions?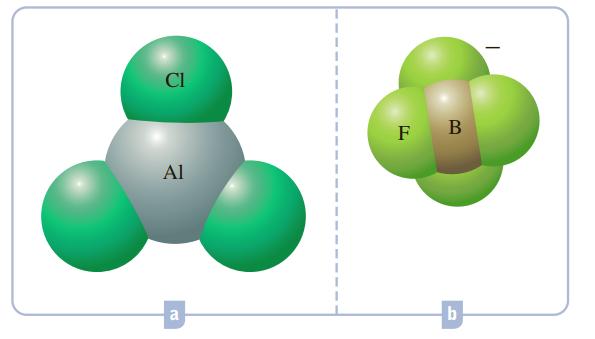
Transcribed Image Text:
CI F B Al a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
a AlCl 3 is a trigonal planar ...View the full answer

Answered By

Dorcas Juliet
I am a proficient tutor and writer with over 4 years experience, I can deliver A+ works in all fields related to business and economics subject. Kindly hire me for excellent papers
4.70+
10+ Reviews
51+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following molecules or ions of sulfur and oxygen, write a single Lewis structure that obeys the octet rule, and calculate the oxidation numbers and formal charges on all the atoms:...
-
Predict the geometry for the central atom in each of the compounds below: a) NH 3 b) H 3 O + c) BH 4 d) BCl 3 e) BCl 4 f) CCl 4 g) CHCl 3 h) CH 2 Cl 2
-
What F 2 phenotypic segregation ratio would be expected for the cross described in the preceding problem if the dominant allele, C, of the third gene produced a product that completely inhibited the...
-
Let a 0. Solve |x| = 3.
-
Explain how a swaption can be terminated at expiration by either exercising it or settling it in cash. Why are these procedures financially equivalent?
-
A sample of 20 CEOs from the Forbes 500 shows total annual compensations ranging from a minimum of $0.1 million to $62.24 million. The average for these 20 CEOs is $7.946 million. The histogram and...
-
Use the data in the first table of Global Insights 6.1 to calculate U.S. tariff revenues on costume jewelry, glass and glassware, and rubber footwear. Global Insights 6.1 The Welfare Costs of...
-
Identify each of the following items as being associated with (a) An AICPA financial statement audit (b) A PCAOB integrated audit (c) Both sets of audit standards _____ AS 5 _____ Must means a...
-
1.How have we manipulated electricity to get us to this point of target interactions? 2.Describe a bremsstrahlung target interaction. 3.Describe a characteristic target interaction. 4.The majority of...
-
Sam Smithers and his brother, Arthur, were avid outdoors enthusiasts and were also quite entrepreneurial. Thus, they agreed to go into business together selling fishing and recreational boats on a...
-
What hybrid orbitals would be expected for the central atom in each of the following molecules or ions? a. SeI 2 b. NO 3 - c. BeCl 2 d. ClO 4 -
-
What hybrid orbitals would be expected for the central atom in each of the following molecules or ions? Cl F a b.
-
For a binary acid, HY, which factors affect the relative ease with which the acid ionizes?
-
What points does a Resident Advisor for a college apartment should always keep in mind while making an advise?
-
Find the magnitude of the linear momentum for the following cases. (Enter your answers in kg . m/s.) (a) a neutron with mass 1.67 x 10-27 kg, moving with a speed of 4.50 106 m/s kg. m/s (b) a 12.0-g...
-
Agarwal, Bergeron, and Cishek have been in partnership for a number of years. The partners allocate all profits and losses on a 2:3:1 basis, respectively. Recently, each partner has become personally...
-
5.71 When a crate with mass 25.0 kg is placed on a ramp that is inclined at an angle a below the horizontal, it slides down the ramp with an acceleration of 4.9 m/s. The ramp is not frictionless. To...
-
______ analysis is a specific type of data analysis that focuses on the composition of the basket, or bundle, of products purchased during a single shopping occasion.
-
Survey results are often reported in newspapers, the evening news, magazines, and even on Facebook! Consider a recent survey reported (about any topic!). Should you accept the results of the survey...
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
a. Draw Lewis structures of each of the following compounds: LiH, NH3, CH4, CO2. b. Which of these has the highest boiling point? Why? c. Which of these has the lowest boiling point? Why? d. Which of...
-
Describe the contents of a carbon dioxide fire extinguisher at 20C. Then describe it at 35C. Explain the difference.
-
Discuss why supercritical carbon dioxide is a nearly ideal solvent.
-
Golden Gate Mining Co. (GGMC) currently has a gold mine operating in Canada and is looking to purchase another gold mine in Peru, operating as Inca Gold Inc. (ICI). You currently work for the CFO at...
-
Cokolo Inc. has entered into the following two derivatives contracts: Purchased put options on 1,000 Trenton Co. shares that have an exercise price of $52 per share and expire in 60 days Negotiated a...
-
As a financial manager (consultant), undertake an investigation on your allocated company to evaluate its strategic financial position. Prepare a business report for the board of directors of the...

Study smarter with the SolutionInn App


