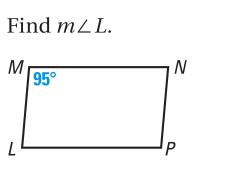
Find the measure of the indicated angle in the parallelogram. Find mLL. M L 95 N P
Question:
Find the measure of the indicated angle in the parallelogram.
Transcribed Image Text:
Find mLL. M L 95° N P
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
In a parallelogram opposite angles are equal Therefore if we know the me...View the full answer

Answered By

Ma Kristhia Mae Fuerte
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Holt McDougal Larson Geometry
ISBN: 9780547315171
1st Edition
Authors: Ron Larson, Laurie Boswell, Timothy D. Kanold, Lee Stiff
Question Posted:
Students also viewed these Mathematics questions
-
The measure of one interior angle of a parallelogram is 50 degrees more than 4 times the measure of another angle. Find the measure of each angle.
-
The measure of one interior angle of a parallelogram is 0.25 times the measure of another angle. Find the measure of each angle.
-
To ensure effective treatment of pinworm infections, which instruction would be most important to emphasize to the patient and family? a. keeping nails long so cutting will not introduce more...
-
Jobs A, B, and C are waiting to be started on Machine Center X. When they are finished at this center, they must be moved to Machine Center Y for final processing. Machine capacity for both centers...
-
Audio Products, Inc., produces two AM/ FM/ CD players for cars. The radio/ CD units are identical, but the mounting hardware and finish trim differ. The standard model fits inter-mediate and full-...
-
Consider a vocabulary with the following symbols: Occupation(p, o): Predicate. Person p has occupation o. Customer (p1, p2): Predicate. Person p1 is a customer of person p2. Boss(p1, p2): Predicate....
-
Consider the following cash flow profile, and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically...
-
Edge Companys production vice president believes keeping up-to-date with technological changes is what makes the company successful and feels that a machine introduced recently would fill an...
-
23 Consider the following Java code: What will be printed when the following code is executed: new Cat().eat(); ? public class Animal ( } public void eat () { System.out.println("Animal is eating");...
-
Determine whether the quadrilateral is a trapezoid. Explain. A D B J C
-
For what value of x is the quadrilateral a parallelogram? 6x 3x + 2
-
Sn and Pb have T c = 3.7 K and 7.2 K respectively. When illuminated with far-IR radiation, they show a marked drop in reflectivity at frequencies of ~10 and ~20 cm 1 , respectively. Rationalize these...
-
Name the following compound: PBr3 Phosphorous bromide Potassium bromide Phosphorous (III) bromide Phosphorous tribromide Potassium(III) bromide Potassium tribromide 0 0 0 0 0
-
When 0.560 g of NA reacts with excess f2 to form NAf 12.8 kk of heat is evolved at standard state conditions
-
In a laboratory setting, concentrations for solutions are measured in molarity, which is the number of moles per liter ( mol / L ) . Concentrations are often converted to more common units on the...
-
nitrogen - 1 3 undergoing positron emission Express your answer as a nuclear equation.
-
A certain liquid X has a normal freezing point of 4 . 2 0 \ deg C and a freezing point depression constant = Kf 4 . 9 7 \ deg C kgmol 1 . A solution is prepared by dissolving some ammonium sulfate (...
-
The following frequency distribution summarizes the weights of 195 fish caught by anglers participating in a professional bass fishing tournament. Weight ( Pounds) Frequency 1 3.......... 53 4...
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Consider the following computer output. (a) Fill in the missing quantities. You may use bounds for the P-values. (b) What conclusions can you draw about the significance of regression? (c) What...
-
Consider the regression model fit to the arsenic data in Exercise 12-16. Use arsenic in nails as the response and age, drink use, and cook use as the regressors. (a) Test for significance of...
-
Consider the regression model fit to the X-ray inspection data in Exercise 12-15. Use rads as the response. (a) Test for significance of regression using = 0.05. What is the P-value for this test?...
-
Four resistors (R = 120 Ohms, R2 = 60 Ohms, R3 = 330 Ohms, and R4 = 480 Ohms), an ideal inductor (L = 230 mH), and a capacitor (C = 250 microF) are connected to a battery (V = 9 V) through a switch...
-
Does International Law exist? Do we have enough by way law making, law enforcement and adjudication at the international level to support the notion that We have a international legal system? their...
-
Jaxons has total revenue of $418,300, earnings before interest and taxes of $102,600, depreciation of $59,200, and a tax rate of 30 percent. The firm is all-equity financed with 15,000 shares...

Study smarter with the SolutionInn App


