(a) Use a molecular orbital program or input and output from software supplied by your instructor to...
Question:
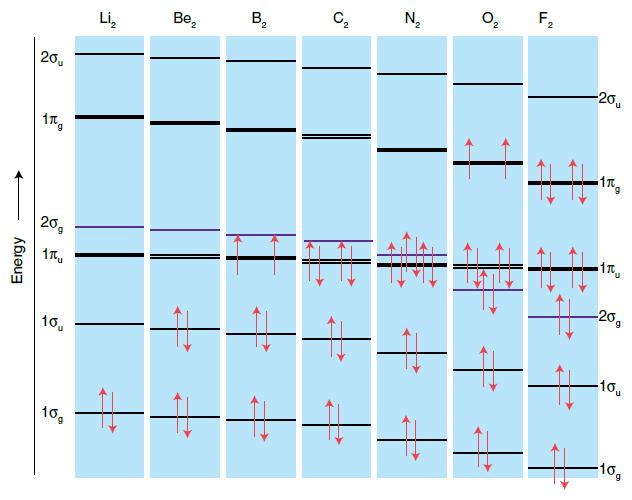
(a) Use a molecular orbital program or input and output from software supplied by your instructor to construct a molecular orbital energy-level diagram to correlate the MO (from the output) and AO (from the input) energies and indicate the occupancy of the MOs (in the manner of Fig. 2.17) for one of the following molecules: HF (bond length 92 pm), HCl (127 pm), or CS (153 pm).
(b) Use the output to sketch the form of the occupied orbitals, showing the signs of the AO lobes by shading and their amplitudes by means of size of the orbital.
Figure 2.17.
Step by Step Answer:
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:




