Answer the following questions using the Frost diagram in Fig. 6.20. (a) What are the consequences of
Question:
Answer the following questions using the Frost diagram in Fig. 6.20.
(a) What are the consequences of dissolving Cl2 in aqueous basic solution?
(b) What are the consequences of dissolving Cl2 in aqueous acid?
(c) Is the failure of HClO2 to disproportionate in aqueous solution a thermodynamic or a kinetic phenomenon?
Figure 6.20.
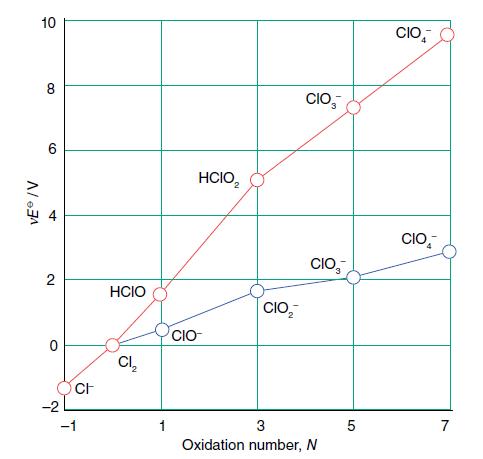
Transcribed Image Text:
VEⓇ /V 10 8 CO 4 2 0 ~ CH -1 HCIO Cl₂ 1 CIO HCIO₂ CIO, CIO, CIO 3 Oxidation number, N сл CIO CIO 7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
A If Cl 2 is dissolved in an aqueous basic solution the anion at the curve is obtained which is an intermediate stage of Cl 2 When all points are conn...View the full answer

Answered By

Vishal madan
Experienced in Science is now likely available on social media here, only for you guys to explore my thoughts, thinking, opinions, and much more to guide humanity. My name is Hammad Shaukat. My greatest passion in life is teaching. I was born and raised in Pakistan (Rawalpindi) and experienced great success at school and in my higher education due to amazing and unforgettable teachers. This is the foundation of my commitment to helping my students, whatever their abilities may be. Currently, I am studying for a bachelor's degree specializing in physics. I have been tutoring and teaching for 2 years in various settings: tutoring small and large groups, private individual tutoring, and teaching in rural, suburban, and urban classroom and home settings. I am specializing in physics. I have much experience in science and am interested in astrology and cosmology. For instructors and tutors, you can mail at any time. ( SPECIALIZED IN SCIENCES ) Major subjects are sciences.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Using the IS-LM and AS-AD to analyze economic shock of the pandemic in March of 2020. a. During March 2020, many states went into a lockdown and most people stopped shopping for goods and services...
-
Thirteen-year-old Matties parents are warm, firm in their expectations, and consistent in monitoring her activities. At school, Mattie met some girls who want her to tell her parents shes going to a...
-
Answer the following questions using the accompanying SPSS output, which is based on data on the growth rates (Y) of depleted chicks at different (log) dosage levels (X) of vitamin B for males and...
-
Voguish Couches is a small company which makes sofas. The company is owned by the couple Lennox and Stewart. You are an intern working at Voguish Couches. For the sake of simplicity, this assignment...
-
Supply the missing information in the following table for Blair Company. Sales ........... $484,000 ROI .......... ? Operating assets ...... ? Operating income ..... ? Turnover ......... 2.2 Residual...
-
Describe the major differences between debt and equity financing.
-
At 3:00 a.m. on November 22, 2010, 16-year-old Sydney McLemore was driving a Mazda3 with her friend, Natalie Hurst, in the front passenger seat. The vehicle was traveling south on Ross Bridge Highway...
-
Prepare a Statement of Activities for Tazewell County for calendar year 20X9, given the following: General property tax revenues . . . . . . . . . . . . . . . . . . . . . . . . . . . $70,000,000...
-
Fine Equipment uses a perpetual inventory system and is located in Vancouver, British Columbia, where the PST rate is 7 % . Fine Equipment uses the earnings approach for revenue recognition. The...
-
Use the data given in Tables 11.1 and 11.3 to calculate the enthalpies of formation for the Group 1 fluorides and chlorides. Plot the data and comment on the trends observed. Table 11.1. Table 11.3....
-
Use standard potential data to suggest why permanganate (MnO 4 ) is not a suitable oxidizing agent for the quantitative estimation of Fe 2+ in the presence of HCl but becomes so if sufficient Mn 2+...
-
Discuss some of the advantages and disadvantages of branded entertainment and give examples of both good and bad uses of this medium.
-
How do postcolonial literary theories critique colonial legacies, cultural imperialism, and the representation of the Other in literature, foregrounding marginalized voices and challenging...
-
PART B Create a table "EMP_StudentID" as follows for Part B. Note: You can store the Date of Joining in any format. EMPID Name PROVINCE Date of Joining Salary 1 JOHN Nova Scotia 9 March, 1998 1000 2...
-
Suppose you already implemented a double hash table class. Running the following code to get the value of a key would return an error. 123 456 7 8 9 def get(self, key): hash1 = hash1fun (key) hash2...
-
a. Use the Product Rule to find the derivative of the given function. b. Find the derivative by expanding the product first. f(x)=(x-6)(x+4)
-
Use the selected balance sheet and income statement information below for Anka Inc. to compute the current ratio. Explain what information this ratio provides. Current Assets Pretax Income Interest...
-
Consider the following unbalanced reaction: P 4 (s) + F 2 (g) PF 3 (g) How many grams of F 2 are needed to produce 120g of PF 3 if the reaction has a 78.1% yield?
-
Write the binomial probability in words. Then, use a continuity correction to convert the binomial probability to a normal distribution probability. P(x 110)
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. The 31 P{ 1 H} NMR spectrum of a CDCl 3 solution of the square planar rhodium(I) complex 4.22 exhibits a doublet of...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. MeCN solutions of NbCl 5 and HF contain a mixture of octahedral [NbF 6 ] , [NbF 5 Cl] , [NbF 4 Cl 2 ] , [NbF 3 Cl...
-
In problems 4.28 to 4.51, refer to Table 4.3 for isotopic abundances where needed. Rationalize the fact that at 173 K, 1 H NMR spectroscopy shows that SbMe 5 possesses only one type of Me group....
-
Research the product Mayonnaise from an economic standpoint (include supply and demand, cost). Describe what has been happening in the product market over the last year or two. Find a balance between...
-
Emilia is Iago's wife, and Desdemona's maid. She's a woman of practical intelligence and emotional resilience. She obeys Iago in a wifely duty, but during the play she develops a strong bond and...
-
Hi, I am doing a business plan for the Centers for Disease Control and Prevention. I need help with Include at least one of the financial statements available from the selected organization (income...

Study smarter with the SolutionInn App


