Assume that the MO diagram of IBr is analogous to that of ICl (Fig. 2.24). (a) What
Question:
Assume that the MO diagram of IBr is analogous to that of ICl (Fig. 2.24).
(a) What basis set of atomic orbitals would be used to generate the IBr molecular orbitals?
(b) Calculate the bond order of IBr.
Figure 2.24
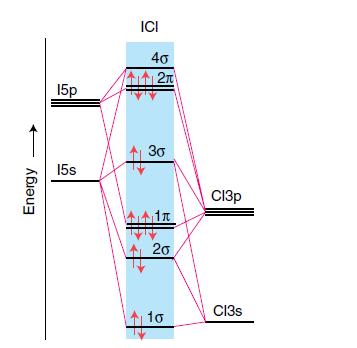
Transcribed Image Text:
Energy 150 15s ICI 40 2π 3G 1л 20 10 СІЗр Cl3s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a To generate the molecular orbitals MOs of IBr the basis set of atomic orbitals used would typicall...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The iodine bromide molecule, IBr, is an inter-halogen compound. Assume that the molecular orbitals of IBr are analogous to the homo nuclear diatomic molecule F2. (a) Which valence atomic orbitals of...
-
The diatomic molecule OH exists in the gas phase. OH plays an important part in combustion reactions and is a reactive oxidizing agent in polluted air. The bond length and bond energy have been...
-
The energy-level diagram in Figure 9.36 shows that the sideways overlap of a pair of p orbitals produces two molecular orbitals, one bonding and one anti-bonding. In ethylene there is a pair of...
-
What was the action you are most proud of, or that is the most meaningful to you? What was the civic issue that was involved in your action? When and how did you first hear about that issue? Why did...
-
Feeback Corporation stock currently sells for $64 per share. The market requires a return of 11 percent on the firms stock. If the company maintains a constant 4.5 percent growth rate in dividends,...
-
In a sequence of sedimentary rock layers, the youngest layer is found at the bottom and the oldest layer at the top. What does this type of layering signify?
-
Kronenberger Burgoyne, LLP, was a law firm with two equity partners who agreed to equal ownership as of 2009. Before 2009, Kronenberger had owned a majority interest in the firm, and when, in 2011,...
-
Grunewald Industries sells on terms of 2/10, net 40. Gross sales last year were $4,562,500, and accounts receivable averaged $437,500. Half of Grunewalds customers paid on the 10th day and took...
-
Medicare is financed through which three primary sources? Public donations, beneficiary premiums, general tax revenue General tax revenue, beneficiary premiums, payroll tax contributions Payroll tax...
-
Assign the lines in the UV photoelectron spectrum of CO shown in Fig. 2.31 and predict the appearance of the UV photoelectron spectrum of the SO molecule. Figure 2.31. 11 13 30 15 // eV 17 1 20 19
-
Draw a molecular orbital energy-level diagram for the gaseous heteronuclear diatomic molecule boron nitride, BN. How does it differ from that for C 2 ?
-
The graph of a normal curve is given. Use the graph to identify the values of and . -1 1 3 5 7 9 11 X
-
What is the knowledge of best practices for Arroyo Fresco Community Health Center, with deadlines and competition? I want to focus on the new strategies for this hospital as transparency, diversity...
-
Last year The Green Grocery had net fixed assets of $48,700. This year, The Green Grocery has net fixed assets of $51,400. Depreciation for this year is $6,300. Which one of the following statements...
-
On December 31, 2019, Munchkin Company had a $367,650 balance in Accounts Receivable. During the year 2020, the company collected $600,000 from its credit customers. The December 31, 2020 balance of...
-
Bill Jr. works on the line at Cloverleaf Automotive just like his fatherpopularly known among the old-timers as Bill Sr.before him. But recently Bill Jr., along with all his coworkers, received...
-
You plan to set up a trust that will provide your newborn son with $1,000,000 on the day he turns 2 If you can earn a 6.5% annual return, how much do you have to deposit in the trust on his 1st...
-
A philanthropic organization knows that its donors have an average age near 60 and is considering taking out an ad in the American Association of Retired People (AARP) magazine. An analyst wonders...
-
A new car sold for $31,000. If the vehicle loses 15% of its value each year, how much will it be worth after 10 years?
-
Comment on each of the following: (a) The difference between extrinsic and intrinsic defects; (b) why CaO is added to ZrO 2 used in refractory materials; (c) The formation of solid solutions of Al 2...
-
Suggest why doping NiO with Li 2 O in air (or the presence of O 2 ) leads to an increase in electrical conductivity, and comment on the dependence of this increase on the amount of lithium dopant.
-
Why are d-block metal oxides much more frequently non-stoichiometric than are non-d-block metal oxides?
-
EXECUTIVE SUMMARY You are a Business Analyst at Simcoe Skies, a regional airline with operations across Eastern Canada (note: this is a fictitious company). Simcoe Skies has received numerous...
-
You are the Manager for a local 24 hour pharmacy store. You hired Maria about two months ago as the Night Shift Lead. Overall her performance has been good. She has a good manner with customers, and...
-
HR Metrics (3-4 pgs with in-text citations (3-4) please): How are employers measuring the effectiveness of their retention strategies? In addition to actual retention statistics and comparison to...

Study smarter with the SolutionInn App


