BINAP is a chelating diphosphine ligand shown below. Discuss the reasons for the observed chirality of BINAP,
Question:
BINAP is a chelating diphosphine ligand shown below. Discuss the reasons for the observed chirality of BINAP, and of its complexes.
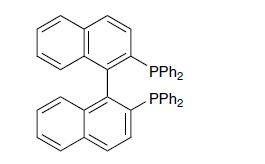
Transcribed Image Text:
PPh₂ PPh₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
BINAP which stands for 22bisdiphenylphosphino11binaphthyl is a chelating diphosphine ligand commonly used in asymmetric catalysis due to its unique ch...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The chiral BINAP ligand shown in Figure 8-7 contains no asymmetric carbon atoms. Explain how this ligand is chiral.
-
Discuss the reasons for and against audio recording an interview.
-
Discuss the reasons for EC failures.
-
Ford Company's cost of capital is 13%. It has invested x (million dollars) in current assets. The following function represents the shortage cost of its current assets: S = 9 e- x/3 Find the...
-
Your pharmaceutical firm is seeking to open up new international markets by partnering with various local distributors. The different distributors within a country are stronger with different market...
-
Use the ogive to approximate (a) the number in the sample. (b) the location of the greatest increase in frequency. Adult Males 80 70 60 50 40 30 20 10 64 66 68 70 72 74 76 Height (in inches) Cu...
-
For Problem 6.8, (a) Fit the corresponding negative binomial model with the same linear predictor. (b) Compare the analysis between part (a) and that from Problem 6.8. 6.8 For the Sexual Health pilot...
-
The Tycron Company produces three electrical products clocks, radios, and toasters. These products have the following resource requirements: The manufacturer has a daily production budget of $2000...
-
As a successful entrepreneur and CEO, you are considering making some financial decisions for your engineering company. Examining the financial report of your company, you notice: . The upgrade of...
-
Sketch the two structures that describe most five-coordinate complexes. Label the two different sites in each structure.
-
The equilibrium constants for the successive reactions of 1,2-diaminoethane with Co 2+ , Ni 2+ , and Cu 2+ are as follows: Discuss whether these data support the generalizations in the text about...
-
A police department has set up a speed enforcement zone on a straight length of highway. A patrol car is parked parallel to the zone, 200feet from one end and 150feet from the other end (see figure)....
-
Suppose an investor in with a 35% tax rate is considering buying a municipal bond with a yield of 3.38%. What is the taxable equivalent yield on the municipal bond? (If your answer is 5.25%, enter...
-
1 You will create JavaScript code - embedded on an HTML file - according to the requirements listed on the next pages. 2 Upload and submit a single html file (.html extension) containing the code...
-
3- Compare and contrast domestic and Eurocurrency money markets. 4- Discuss the systematic risk of a foreign project compared with a purely domestic investment. Explain how to estimate the...
-
Find the truth table that describes the following circuit: YO 20 D D D OF
-
Alina deposited $33,000 with a bank in a five-year certificate of deposit kneeling 5% interest compounded daily. Find the interest earned on the investment. Use the table below with a compound...
-
An experimental fuel cell has been designed that uses carbon monoxide as fuel. The overall reaction is 2CO(g) + O2(g) 2CO2(g) The two half cell reactions are CO + O2- CO2 + 2e- O2 + 4e 2O2- The...
-
Which of the following is FALSE regarding the purchasing power parity (PPP). a. The PPP is a manifestation of the law of one price b. The PPP says that a country with a higher expected inflation can...
-
Treatment of (2,4,6- t Bu 3 C 6 H 2 )P=P(2,4,6- t Bu 3 C 6 H 2 ) with CF 3 SO 3 Me gives a salt A as the only product. The 31 P NMR spectrum of the precursor contains a singlet (+495 ppm), while that...
-
(a) In 1956, it was concluded on the basis of dipole moment measurements that Cp 2 Pb did not contain coparallel C 5 -rings. Explain how this conclusion follows from such measurements. (b) X-ray...
-
Rationalize the following observations. (a) On forming the CC bond in C 2 (CN) 4 lengthens from 135 to 151 pm. (b) During the photolysis of Mo(CO) 5 (THF) with PPh 3 , a signal in the 31P NMR...
-
4. Does the following graph G have a perfect matching? explain
-
Write a computer program to compute the A, B and D matrices for an arbitrarily stacked laminate having layers of different but uniform thickness. Have an option to have some layers made of a...
-
3. State the clique-number for the following graph G. a b e ENZ d 9 h

Study smarter with the SolutionInn App


