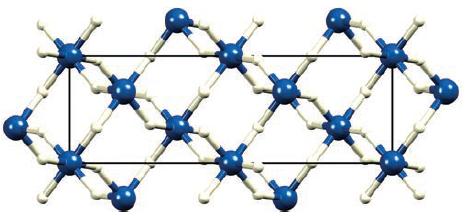
Confirm that the 3D structure of aluminium hydride shown in Fig. 10.17 is consistent with a 1
Question:
Confirm that the 3D structure of aluminium hydride shown in Fig. 10.17 is consistent with a 1 : 3 (Al : H) stoichiometry.
Figure 10.17
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
In Figure 1017 the 3D structure of aluminum hydride AlH3 is ...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. The Incisors own a rental beach house in Hawaii. The beach house was rented for the full year during 2012...
-
A rotating object has an angular acceleration of = 0 rad/s2. Which one or more of the following three statements is consistent with a zero angular acceleration? A. The angular velocity is w = 0...
-
Featherstone Inc. reported the following data: Net income ................................... $296,000 Depreciation expense ................... 113,100 Gain on disposal of equipment ...... 58,500...
-
Valley View Hospital has an outpatient clinic. Jeffrey Harper, the hospitals chief administrator, is very concerned about cost control and has asked that performance reports be prepared that compare...
-
Following are several transactions involving a university. 1. In the current fiscal year, the university was notified by the federal government that next fiscal year it would receive a $500,000 grant...
-
Why does collusion pose unique prevention and detection challenges?
-
The popularity of Southwestern Universitys football program under its new coach, Bo Pitterno, surged in each of the 5 years since his arrival at the Stephenville, Texas, college, (Southwestern...
-
Ruler Formula Bar Normal Page Break Page Custom Preview Layout Views Gridlines Headings Zoom 100% Zoom To Selection Window Macros Workbook Views Show Zoom Macros A A1 V A B D E F G H 4 Assume That...
-
(a) SbCl 3 may be used as a non-aqueous solvent above its melting point. Suggest a possible self-ionization process for this solvent. (b) Explain why the reaction of NOCl with AgNO 3 in liquid N 2 O...
-
Solutions of H 2 O 2 are used as bleaching agents. For the decomposition of H 2 O 2 to H 2 O and O 2 , Why can H 2 O 2 be stored for periods of time without significant decomposition? AG -116.7kJ mol
-
Do firms have any responsibilities to society at large? MINI CASE Assume that you recently graduated with a degree in finance and have just reported to work as an investment advisor at the brokerage...
-
Gwendolyn Berry pled guilty to stealing funds from her employer. At sentencing, she was ordered to pay restitution of more than $2 million. To enforce this judgment, the government garnished 50...
-
An insurance applicant is usually protected from the time an application is made, if a premium has been paid, possibly subject to certain conditions. (True/False)
-
The board of directors of U.S. Goods Corporation announces that the corporation will pay a cash dividend to its shareholders. Once declared, a cash dividend is a. a corporate debt. b. a personal debt...
-
An appraiser specializes in estimating property values. (True/False)
-
A writ of execution is issued before the entry of a final judgment. (True/False)
-
If the sales of a foreign subsidiary all occurred on one day during the year, would the sales be translated at the average rate for the year or the rate on the date of the sales? Explain.
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
The molecular geometries of crown ether derivatives play an important role in capturing and transporting alkali metal ions. K. Okano and co-workers (see K. Okano, H. Tsukube, and K. Hori,...
-
Identify the metal-containing compounds A, B, C, and D in this scheme: Ba- HO - A HO D CO carbon B
-
Account for the fact that LiF and CsI have low solubility in water whereas LiI and CsF are very soluble.
-
How do microbial growth kinetics, characterized by phases such as lag, exponential, stationary, and death phases, provide insights into the dynamic interplay between microbial physiology,...
-
How well do you think employee surveys would do for a company looking to improve?
-
Common stock value-Constant growth McCracken Roofing, Inc., common stock paid a dividend of $1.11 per share last year. The company expects earnings and dividends to grow at a rate of 4% per year for...

Study smarter with the SolutionInn App


