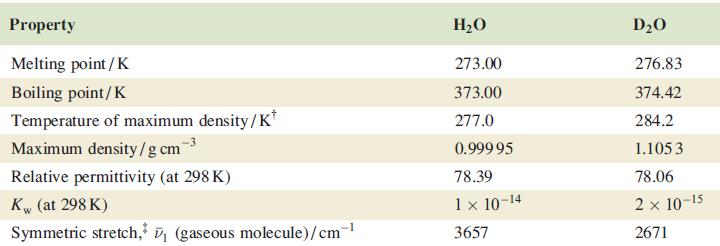
Confirm that the difference in values of (OH) and (OD) given in Table 10.2 is consistent with
Question:
Confirm that the difference in values of ν̅(O—H) and ν̅(O—D) given in Table 10.2 is consistent with the isotopic masses of H and D.
Table 10.2
Transcribed Image Text:
Property Melting point / K Boiling point/K Temperature of maximum density/K* Maximum density/gcm Relative permittivity (at 298 K) Kw (at 298 K) Symmetric stretch, ₁ (gaseous molecule)/cm-¹ H₂O 273.00 373.00 277.0 0.999 95 78.39 1 × 10-14 3657 D₂O 276.83 374.42 284.2 1.105 3 78.06 2 x 10-15 2671
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To confirm whether the difference in values of OH and OD given in Table 102 is consistent with the i...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
ABC Inc. is a U.S. manufacturing firm. It is currently in the process of issuing 20-year bonds with face value of $10,000. They expect the bond to have a credit rating of "A", and comparable A-rated...
-
In Chapter 20, we learned that to compute an approximate 90% confidence interval, the appropriate multiplier is 1.645 instead of 2.0. This works for confidence intervals for one mean or the...
-
The motivation for getting the MBA degree has many aspects-the prestige, greater opportunity for promotion, change of occupation, and an increase in pay. To focus just on this last motivation,...
-
State Newtons second law of motion. What are the limitations on the use of Newtons second law? Explain.
-
Wilmington Composites, Inc., developed its overhead application rate from the annual budget. The budget is based on an expected total output of 720,000 units requiring 3,600,000 machine hours. The...
-
The Shannon Community Kitchen provides hot meals to homeless and low-income individuals and families; it is the organizations only program. It is the policy of the community kitchen to use restricted...
-
Gilead is a large drug producer, with a majority of its prescription drug product sales occurring in the United States. Gilead produces anti-HIV drug therapies, including the drugs Atripla, Truvada,...
-
Shine King Cleaning has decided that, in addition to providing cleaning services, it will sell cleaning products. During December, Shine King completed the following transactions: Dec 2 Purchased 600...
-
please explain this answer by Relational model drawing Question # 01: Convert the given ER model into Relational Model Fname Minit Lname Bdate Name Address Salary Sen Sex Locations WORKS FOR (4,N)...
-
Using the information in Figs. 10.14 and 10.18, explain how the two oligonucleotides assemble into a double helical structure (see Fig. 10.14 for the 3' and 5' numbering, and definitions of C, A, G...
-
Ionic liquids show potential for applications in lithium-ion batteries. A combination of Li[N(SO 2 CF 3 ) 2 ] and [EMIm][N(SO 2 CF 3 ) 2 ] (EMim = 1-ethyl-3 methylimidazolium ion) has been used as a...
-
The average cost of a speeding ticket plus court fees is approximately $150. A random sample of 38 speeding ticket court cases showed that the mean cost was $152.59. At the 0.01 level of...
-
Jeremy incorporated FormFit Concrete, Inc., but did not file its first annual report, so the state involuntarily dissolved the firm. Unaware of this, Jeremy contracted with Market Square to lay the...
-
Where are bankruptcy proceedings held?
-
What is the floating lien concept?
-
Jill is a shareholder of United Manufacturing Company. As a shareholder, Jills rights include all of the following except a right to a. have one vote per share. b. access corporate books and records....
-
Ishana files a Chapter 7 petition for a discharge in bankruptcy. Ishana may be denied a discharge on which of the following grounds? a. Concealing property with the intent to defraud a creditor. b....
-
The application of the lower of cost and net realizable value requirement to the translated financial statements requires different treatment with regard to the two classifications of foreign...
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
Discuss the industrial uses of lithium and likely future demand for compounds of the metal. How are these demands likely to be met? A useful resource is the United States Geological Survey at...
-
Under ambient conditions lithium and sodium adopt simple bcc structures. Under high pressures these alkali metals undergo a series of complex phase transitions to fcc and then lowersymmetry...
-
The auride anion, Au , has a similar ionic radius to Br at 196 pm. Predict structures for the ionic compounds CsAu and RbAu.
-
Item Prior year Current year Accounts payable 8,141.00 7,967.00 Accounts receivable 6,027.00 6,590.00 Accruals Cash 1,043.00 1,630.00 ??? ??? Common Stock COGS Current portion long-term debt...
-
At 3 1 December 2 0 1 1 Donna had receivables totaling $ 2 5 0 , 0 0 0 and an allowance for receivables of S 2 4 , 0 0 0 brought forward from the previous year. It has been decided to write off...
-
what ways do microbial growth models, including mathematical models, agent-based simulations, and genome-scale metabolic reconstructions, facilitate the prediction and optimization of microbial...

Study smarter with the SolutionInn App


