Describe the bonding in Ga 2 H 6 and Ga 2 Cl 6 , both of which
Question:
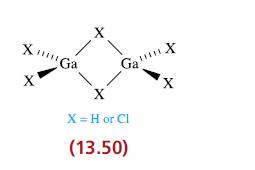
Describe the bonding in Ga2H6 and Ga2Cl6, both of which have structures of the type shown in 13.50.
Transcribed Image Text:
XmGa X Ga X X = H or Cl (13.50) X
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The compounds Ga2H6 digallane and Ga2Cl6 digallium hexachloride both exhibit unique bonding patterns ...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Describe the bonding in the CO32- ion using the LE model. How would the molecular orbital model describe the bonding in this species?
-
Describe the bonding in the O3 molecule and the NO2- ion using the LE model. How would the molecular orbital model describe the bonding in these two species?
-
The space-filling models of ethane and ethanol are shown below. Use the LE model to describe the bonding in ethane and ethanol. Ethane Ethanol (C2Ha)CH,OH) 0
-
The two roots of a quadratic equation ax 2 + bx + c = 0 can be obtained using the following formula: b 2 - 4ac is called the discriminant of the quadratic equation. If it is positive, the equation...
-
Explain how a gain or loss on disposal is handled in a capital-budgeting analysis.
-
As we see in Exercise 10.44, or by using the data in StudentSurvey, the regression line to predict Weight from Height is Figure 10.8 shows three graphs for this linear model: the scatterplot with...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/ year. a. Determine the MIRR for this project. b. Is this project economically...
-
Use the following information to prepare a classified balance sheet for Alpha Co. at the end of 2016. Accounts receivable .....$26,500 Accounts payable ....... 12,200 Cash ............ 20,500 Common...
-
The following are the ages of 13 mathematics teachers in a school district. 28, 30, 34, 34, 36, 38, 39, 42, 46, 47, 49, 50, 51 Notice that the ages are ordered from least to greatest. Give the...
-
Suggest explanations for the following facts. (a) Na[BH 4 ] is very much less rapidly hydrolysed by H 2 O than is Na[AlH 4 ]. (b) The rate of hydrolysis of B 2 H 6 by water vapour is given by the...
-
(a) One gallium-containing product, A, was obtained from the following reaction, carried out in Et 2 O solvent: The room temperature, solution 1 H NMR spectrum of A showed the following signals: ...
-
Each of the following situations occurred during 2024 for one of your audit clients: 1. An inventory write-down due to obsolescence. 2. Discovery that depreciation expenses were omitted by accident...
-
What are the major sources of uncertainty in an environment? Discuss how these sources of uncertainty affect a small biotechnology company and a large carmaker.
-
Ask a manager to describe both an instance of ethical behavior and an instance of unethical behavior that she or he observed. What caused these behaviors, and what were the outcomes?
-
What interorganizational strategies might work most successfully as a company expands globally? Why?
-
Under what conditions is an organization likely to prefer (a) a mechanistic structure, (b) an organic structure, or (c) elements of both?
-
Why does differentiation occur in an organization? Distinguish between vertical and horizontal differentiation.
-
Acme Corp. has a portfolio of investments purchased at the amounts shown below at December 31, 2013. Acme is a private company but is contemplating going public. 1. 10% interest in Plato purchased on...
-
A line l passes through the points with coordinates (0, 5) and (6, 7). a. Find the gradient of the line. b. Find an equation of the line in the form ax + by + c = 0.
-
The perovskite structure, ABX 3 , can be described as a closepacked array of the A and X ions together, with B-type cations in octahedral holes. What proportion of octahedral holes is filled?
-
The structure of calcite (CaCO 3 ) is shown in Fig. 4.76. Describe how this structure is related to that of NaCl. Figure 4.76. CO Ca+
-
(a) Calculate the enthalpy of formation of the hypothetical compound KF 2 assuming a CaF 2 structure. Use the BornMayer equation to obtain the lattice enthalpy and estimate the radius of K 2+ by...
-
At January 1, 2023, Trailblazers Corporation had 300,000 common shares outstanding (no preferred issued). On March 1, the corporation issued 500,000 new shares to raise additional capital. On July 1,...
-
what ways do advances in imaging technologies, such as super-resolution microscopy, live-cell imaging, and genome-wide chromosome conformation capture (Hi-C), provide insights into the dynamic...
-
A company's manufacturing proccess requires $18.50 worth of material for every $10 spent on labour. How much should they allocate budget for material if the labour budget is $19,540?

Study smarter with the SolutionInn App


