Determine the g values of the EPR spectrum shown in Fig. 8.57, measured for a frozen sample
Question:
Determine the g values of the EPR spectrum shown in Fig. 8.57, measured for a frozen sample using a microwave frequency of 9.43 GHz.
Figure 8.57
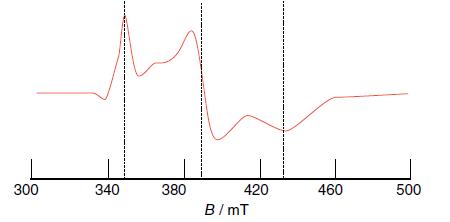
Transcribed Image Text:
300 340 380 420 B/mT 460 500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The gvalues of the EPR spectrum shown in Fig 857 can be determined using the following equation hv g ...View the full answer

Answered By

KIRAN P
I have M TECH in Chemical Engineering from one of India's best Institute NIT TRICHY and have more than 10 years experience in Chemical industry.Used to teach students during leisure times
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A researcher wanted to find out if there was difference between older movie goers and younger movie goers with respect to their estimates of a successful actors income. The researcher first...
-
The following table gives the systolic blood pressure (SBP), body size (QUET), age (AGE), and smoking history (SMK = 0 if a nonsmoker, SMK = 1 if a current or previous smoker) for a hypothetical...
-
Helium gas is throttled steadily from 500 kPa and 70C. Heat is lost from the helium in the amount of 2.5 kJ/kg to the surroundings at 25C and 100 kPa. If the entropy of the helium increases by 0.25...
-
Presented below is financial information related to the 2015 operations of Louisa Cruise Company. Maintenance and repairs expense .......$ 92,000 Utilities expense ............... 10,000 Salaries and...
-
You borrowed $2,000 to take a vacation in the Caribbean islands. At the end of the year, you had to pay back $2,200. What is the annual interest that you paid on your loan?
-
99 percent per month. In 2014, Boling resolved his suit against the gas can manufacturer. Shortly thereafter, Prospect sent Boling a Schedule of Purchases, asserting that Boling owed Prospect...
-
A sample of nine public universities and nine private universities was taken. The total cost for the year (including room and board) and the median SAT score (maximum total is 2400) at each school...
-
A 25-year-old Latin, G1P1, cisfemale presents to the office with a 6-month history of amenorrhea; reports having regular menstrual cycles every month up until about 1 year ago when her menstrual...
-
How would the cyclic voltammetry shown in Fig. 8.53a differ if (a) The Os(IV) complex decomposed rapidly; (b) Os(III) is oxidized in a single, rapid two-electron step to Os(V)? Figure 8.53a. i/mA 10...
-
The solution 31 P-NMR spectrum of P 4 S 3 consists of a doublet and quartet with intensities in the ratio 3:1. Suggest a structure consistent with this pattern.
-
Sumarah Corp. accepted a $450,000 two year note receivable from a customer in connection with a major inventory sale transaction on 1 October 20X5. The note was interest free, although market...
-
Question 3 Discuss how price indicators, like the A-D line, are commonly used to identify major turning points such as the end of a bull market. Question 3 Discuss how price indicators, like the A-D...
-
In the space Rn, do the following sets form linear spaces? x2 ,--()., = = ... | X1, X2, Xn ER, x1 + x2 + + xn = 1 -- = = = .-{-- V3 = 1x1' X2 ..., | X1, X2,...,xn ER, x1 + x2 + + xn = 0 | X1, X2, :...
-
write a program that calculates information about a sequence of tracks on an album. Write a Java main class called YourlastnameProgram6.java (with your actual last name) that does the following:...
-
Why was the 60/40 stock/bond portfolio mix considered a hedge and why is that potentially problematic in todays market?
-
Please find an organisation online to use as a reference. Find one (1) insurance product your company offers, briefly summarise the role of the following terms: '-"Standard cover and deviations...
-
The following is an energy- level diagram illustrating three different electronic transitions in the Bohr hydrogen atom. a. Explain why the energy levels get closer together as they increase. Provide...
-
Charles owns an office building and land that are used in his trade or business. The office building and land were acquired in 1978 for $800,000 and $100,000, respectively. During the current year,...
-
Comment on the modes of bonding of the ligands in the Mn(II) complexes listed at the end of Section 21.8, drawing attention to any conformational restrictions. Data from Section 21.8 Pr Pr iPr Mn iPr...
-
Values of oct for [Ni(OH 2 ) 6 ] 2+ and high-spin [Mn(OH 2 ) 6 ] 3+ have been evaluated spectroscopically as 8500 and 21000 cm 1 respectively. Assuming that these values also hold for the...
-
For which of the following ions would you expect the spin-only formula to give reasonable estimates of the magnetic moment: (a) [Cr(NH 3 ) 6 ] 3+ , (b) [V(OH 2 ) 6 ] 3+ , (c) [CoF 6 ] 3 ? Rationalize...
-
For decades, the Taylor School has been providing elementary education in the sector where it's located. While other institutions in the area have faced difficulties, the school has maintained a good...
-
What are the uses of recombinant DNA pharmaceuticals?
-
on Project Risk Management, and describe how each of the 6 elements of project and portfolio risk management work together. In addition, do you see a need for a seventh element? Briefly explain why....

Study smarter with the SolutionInn App


