Explain why SF 6 is a very stable molecule whereas OF 6 is unknown. Make use of
Question:
Explain why SF6 is a very stable molecule whereas OF6 is unknown. Make use of the bond enthalpy data given in Table 2.7.
Table 2.7.
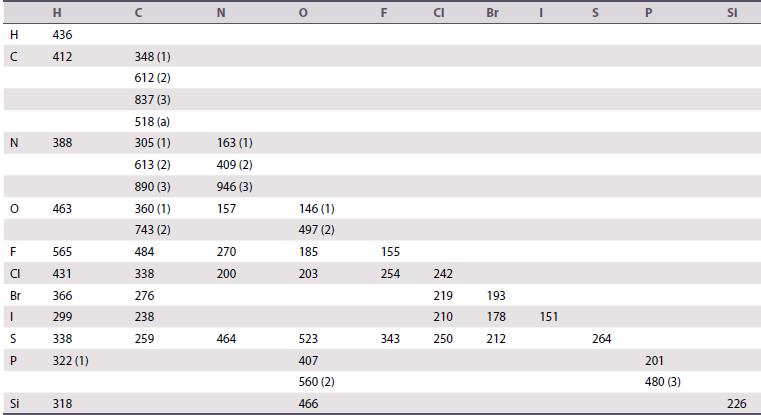
Transcribed Image Text:
H с N 0 F CI Br | S P LL Si H 436 412 388 463 565 431 366 299 338 322 (1) 318 348 (1) 612 (2) 837 (3) 518 (a) 305 (1) 613 (2) 890 (3) 360 (1) 743 (2) 484 338 276 238 259 N 163 409 (2) 946 (3) 157 270 200 (1) 464 146 (1) 497 (2) 185 203 523 407 560 (2) 466 F 155 254 343 P 242 219 210 250 Br 193 178 212 151 S 264 201 480 (3) SI 226
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
To understand why SF6 is a very stable molecule whereas OF6 is unknown we need to compare the bond e...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Main street apartments has 4 apartments rented at $1075/month.Laundry income of $5370 per year and 11 storage lockers rented at $30 per month. The vacancy rate is 1.6%. What is the effective gross...
-
Explain why borazole (sometimes called inorganic benzene) is a very stable compound. N:N borazole
-
In the conversion of a-ketoglutarate to glucose, which of the following compounds are not obligatory intermediates in this pathway? * Oxaloacetate O Fructose 1,6 bisphosphate O Malate O 1,3...
-
Route 66 Tire Co. manufactures automobile tires. Standard costs and actual costs for direct materials, direct labor, and factory overhead incurred for the manufacture of 10,000 tires were as follows:...
-
What is the distinction between repeated measurements and matched items?
-
In question 1, identify the marketing as opposed to the business strategy. Data From Question 1: What is a business strategy? Do you agree with the definition proposed? Illustrate your answer with...
-
You have signed a contract to build a garage for the Simpsons. You will receive a $500 bonus for completing the project within 15 working days. The contract also contains a penalty clause in which...
-
The given code snippet is implemented using linear probing technique, what change you have to make so that the quadratic probing is implemented. @Override public boolean put(String word, String...
-
State whether the following oxides are acidic, basic, neutral, or amphoteric: CO 2 , P 2 O 5 , SO 3 , MgO, K 2 O, Al 2 O 3 , CO.
-
Sketch the two possible geometric isomers of the octahedral [AsF 4 Cl 2 ] and explain how they could be distinguished by 19 F-NMR.
-
How does Markel Corporation gain and lose from fluctuating exchange rates?
-
Housekeeping Services at Omega Hospital provided janitorial services to three wards during 2014 at a direct cost of $100,000. The square footage of each ward and hours of housekeeping are provided...
-
Cow-Calf Budget Spring Calving, Sell at 225 days of age Feeder Calf Sale $340 Total revenue Income over variable costs $30999 Profit $30694 Breakeven selling price needed for the feeder calf to pay...
-
Search for cut off/standard ratio for merchandising businesses At least 2 links resources per ratio Profitability a. Net Profit Margin b. Gross Profit Margin c. Operating Profit Margin d. Return on...
-
what is the Nikeinventory reserve? Nike make ongoing estimates relating to the net realizable value of inventories based upon our assumptions about future demand and market conditions. If we estimate...
-
A living will: Question 27 options: Expresses the client's desire to transfer assets to designated heirs. Requires a court to appoint a Guardian for an incompetent person. Expresses the client's...
-
The herb ginkgo Biloba is commonly used as a treatment to prevent dementia. In a study of the effectiveness of this treatment, 1545 elderly subjects were given ginkgo and 1524 elderly subjects were...
-
How is use of the word consistent helpful in fraud reports?
-
Deduce possible J values for a 3 F term. What is the degeneracy of each of these J levels, and what happens when a magnetic field is applied? Sketch an energy level diagram to illustrate your answer,...
-
Which of the following ions are diamagnetic: Rationalize your answer. (a) [Co(OH)6]+, (b) [CoF, (c) [NiF, (d) [Fe(CN)6], (e) [Fe(CN)6], (f) [Mn(OH)6]+?
-
What are the limitations of the RussellSaunders coupling scheme?
-
Particles A and B, of mass 4 kg and 3 kg respectively, are connected together by means of a light inextensible string. Particle A rests on a smooth plane inclined at 40 to the horizontal. The string...
-
Use the differentiability equation: f'(c) = lim x- f(x) f(c) x-c To determine whether or not f(x) = (x 3) (2+x) is differentiable at x = 3 If not, - state the type of non-differentiable point. If so,...
-
A metal wire of mass m, and length L lies in a non-uniform magnetic field that varies with respect to x as given by B(x) = Box/L. If a constant known current i passes through the wire, determine the...

Study smarter with the SolutionInn App


