Predict the standard enthalpies of the reactions by using mean bond enthalpy data. Assume that the unknown
Question:
Predict the standard enthalpies of the reactions
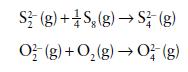
by using mean bond enthalpy data. Assume that the unknown species O42− is a singly bonded chain analogue of S42− .
Transcribed Image Text:
S2(g) +S₂(g) → S2 (g) O2(g) + O₂(g) → 0² (g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a S 2 2 g 14 S 8 g S 4 2 g The enthalpy change for this reaction is zero because there is no c...View the full answer

Answered By

User l_1013947
I possess a comprehensive understanding of programming languages such as C++, Python, HTML, CSS, and Jupyter Notebook. These technical skills enable me to develop robust software solutions and create visually appealing web pages. With my expertise in coding, I can effectively tackle complex programming tasks and deliver high-quality results.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The standard enthalpies of formation of ClO and ClO2 are 101 and 102 kJ/mol, respectively. Using these data and the thermodynamic data in Appendix C, calculate the overall enthalpy change for each...
-
`The standard enthalpies of formation of ions in aqueous solutions are obtained by arbitrarily assigning a value of zero to H+ ions; that is, Hf [H+(aq)] = 0. (a) For the following reaction ...
-
The standard enthalpies of formation of S(g), F(g), SF4(g), and SF6(g) are 1278.8 kJ/ mol, 179.0 kJ/ mol, 775 kJ/ mol, and 1209 kJ/ mol, respectively. a. Use these data to estimate the energy of an...
-
Workplace stress is sometimes related to time management. Describe a time when you were stressed at work or school because of time management. Explain how you would advise an employee or student to...
-
In the chapter, we mentioned that many companies have been under pressure to declassify their boards of directors. Why would investors want a board to be declassified? What are the advantages of a...
-
If the Sun were the size of a beach ball, Earth would be the size of a green pea 110 m away. Show that the nearest star, Alpha Centauri (4.4 light-years away), would be about 30,000 km distant. (Find...
-
Amanda Forbes was hired as a nail technician by Showmann, Inc., in 2011. In 2017, Forbes attended a work-related holiday party where Showmann distributed raffle tickets to employees. One of the...
-
Ratio analysis Required Use the financial statements for Bernard Company from Problem 13-22A to calculate the following ratios for 2012 and 2011: a. Working capital b. Current ratio c. Quick ratio d....
-
Use counting to determine the whole number that corresponds to the cardinality of these sets. (a) C = {xlx = N and (x-8)(x-3)=0} (b) D = {x|x EN, 1x 100 and x is divisible by both 6 and 7} (a) n(C) =...
-
Solid phosphorus pentachloride is an ionic solid composed of PCl 4 + cations and PCl 6 anions, but the vapour is molecular. What are the shapes of the ions in the solid?
-
In which of the species lCl 6 and SF 4 is the bond angle closest to that predicted by the VSEPR model?
-
Why do increases in oil prices lead to increases in food prices? Describe the various factors affected by rising oil prices that, in turn, contribute to rising food prices.
-
How do ethical leaders navigate complex ethical dilemmas while upholding the integrity and reputation of the organization?
-
1. Let an economy be characterized by two people, Jack and Jill. Each period Jack buys 10 fish from Jill and Jill buys 15 coconuts from Jack. a. If the price of fish is $3 and the price of coconuts...
-
If two businesses are operating in excess of their break even sales level and both increase by 15%, what will happen to their profitability?
-
what is the compa-ratio for a salary range of $10 - $22 and an entry-level employee salary of $12.50?
-
What is the Army's standard fielding process developed to provide Army materiel systems to units in a coordinated/consolidated package of end items called?
-
A Gallup Poll (Exercise 57) asked Americans if the fact that they can make copies of songs on the Internet for free made them more likely or less likely to buy a performers CD. Only 13% responded...
-
What services are provided by the provincial and territorial governments?
-
(a) The lists below show wrongly paired molecules or ions and point groups. Assign the correct point group to each species. (b) A molecule X 2 H 6 belongs to the D 3d point group. Does the molecule...
-
What is meant by a ligand group orbital?
-
In the description of the bonding of B 2 H 6 , we draw the conclusion that the two bonding MOs in Fig. 5.33 have BH bonding character delocalized over the four bridge atoms. (a) What other character...
-
What is "Time Study" and what purpose does it serve? What is "Value Analysis"? What is forecasting? Describe the steps in the forecasting process? Describe the various elements in the time series...
-
How would a data analyst go about employing Cluster Analysis as a solution technique? Be very careful to include all steps from the discovery of the need to use Cluster Analysis to the ultimate...
-
Complete the table to identify specific requirements in your area of work and the responsibilities of managers to develop and monitor policies and procedures to meet legal and ethical frameworks....

Study smarter with the SolutionInn App


