Predict whether the equilibrium constants for the following reactions should be greater than 1 or less than
Question:
Predict whether the equilibrium constants for the following reactions should be greater than 1 or less than 1:
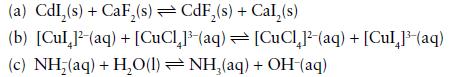
Transcribed Image Text:
(a) CdI₂(s) + CaF₂(s) CdF₂(s) + Cal,(s) (b) [Cul 1¹-(aq) + [CuCl₂]³(aq) (c) NH;(aq) +H,O(l)=NH,(aq) [CuCl²-(aq) + [Cul 1³ (aq) + OH(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To predict whether the equilibrium constants Kc for the given reactions are greater than 1 or less t...View the full answer

Answered By

Deepankur Keserwani
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
(a) Based on the following energy profile, predict whether kf > or kf (b) Using Equation 15.5, predict whether the equilibrium constant for the process is greater than 1 or less than 1. 6r Reactants...
-
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the equation:
-
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the equation:
-
Governmental Funds Statement of Revenues Expenditures and Changes in Fund Balance. You have recently started working as the controller for a small county. The county is preparing its financial...
-
Use the standard price and cost data supplied in Problem 8-20B. Assume that Truebody actually produced and sold 432,000 units. The actual sales price and costs incurred follow. Actual price and...
-
Differentiate among growth, small-cap, mid-cap, dividend, and balanced growth and income funds.
-
An often-ignored concept in breach of contract is the availability, if any, of the award of punitive damages. Often, cases incorporate both breach of contract and tort actions. The tort actions...
-
The beginning inventory of merchandise at Dunne Co. and data on purchases and sales for a three-month period ending June 30, 2014, are as follows: Instructions 1. Record the inventory, purchases, and...
-
What is operating leverage and how is it related to the cost structure of any organisation ?
-
The f-block elements are found as M(III) lithophiles in silicate minerals. What does this indicate about their hardness?
-
Is the dissolution of silicates in HF a Lewis acidbase reaction, a Brnsted acidbase reaction, or both?
-
You are offered the right to receive $1000 per year forever, starting in one year. If your discount rate is 5%, what is this offer worth to you?
-
Mark (67) and Josephine (69) are married and are both U.S. citizens. They will file a joint return. Which federal tax form do they have the option to use to file their personal income tax return?
-
The word 'peacekeeping' is not mentioned in the UN Charter, yet this is one of the most recognizable activities of the United Nations. What is its legal basis?
-
6. (10%) Given a received vector (1,2,..., 6) = (1,-1,-1,-2,-2,1), please use the A* algorithm to find a path from the root of the following code tree to a goal node such that the cost of the path is...
-
For public and mass communication, make an observation how the following communication was delivered. Highlight its differences. News on TV Product Advertising Public Service Radio Broadcasting (no...
-
Bright sunlight is reflected from an icy pond. You look at the glare of the reflected light through polarized glasses. When you take the glasses off, rotate them 90, and look through one of the...
-
The reusable booster rockets of the U.S. space shuttle use a mixture of aluminum and ammonium perchlorate for fuel. A possible equation for this reaction is 3Al(s) + 3NH4ClO4(s) Al2O3(s) + AlCl3(s)...
-
Interview managers at three companies in your area about their use of ERP. How have their experiences been similar? What accounts for the similarities and differences?
-
(a) Give a definition of lattice energy. Does your definition mean that the associated enthalpy of reaction will be positive or negative? (b) Use the BornLande equation to calculate a value for the...
-
(a) Draw a set of resonance structures for the hypothetical molecule PH 5 , ensuring that P obeys the octet rule in each structure. Assume a structure analogous to that of PF 5 . (b) To what point...
-
Refer to Table 6.2. (a) Write an equation for the process for which the standard enthalpy of atomization of cobalt is defined. (b) Suggest reasons for the trend in standard enthalpies of atomization...
-
Case 1: Murfam Enterprises LLC , TCM 2023-73 The summary should include the following five sections: (1) case citation, (2) facts, (3) issue(s), (4) holdings, and (5) rationale.
-
Product C Product J Product R Sales Wariable expenses $330,000 $190,000 $330,000 198,000 133,000 264,000 Contribution margin $132,000 $57,000 $66,000 Total $850,000 595,000 $255,000 1. Margin of...
-
Consider the grammar below and apply the following procedures SA | ACa | AbA A Aa e BBC | BA | bC C Cb | CB (a) Eliminate any productions. (b) Eliminate any unit productions in the resulting grammar....

Study smarter with the SolutionInn App


