Showthat the metal centres in the following complexes obey the 18-electron rule: (a) (n-Cp)Rh(n-CH4) (PMe3) (b) (n-C3H5)2
Question:
Showthat the metal centres in the following complexes obey the 18-electron rule:
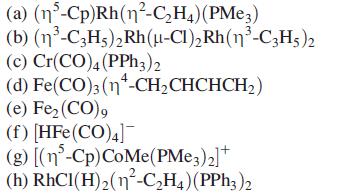
Transcribed Image Text:
(a) (n³-Cp)Rh(n²-C₂H4) (PMe3) (b) (n³-C3H5)2 Rh (μ-Cl)₂ Rh(n³-C3H5) 2 (c) Cr(CO)4 (PPH3)2 (d) Fe(CO)3 (n-CH₂CHCHCH₂) (e) Fe₂ (CO), (f) [HFe (CO)4] (g) [(n-Cp) CoMe (PMe3) 2] + (h) RhCI(H)₂(n²-C₂H4) (PPh3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
EAN outermost ELECTRONoxidation state electron gathered by ligan...View the full answer

Answered By

User l_1013947
I possess a comprehensive understanding of programming languages such as C++, Python, HTML, CSS, and Jupyter Notebook. These technical skills enable me to develop robust software solutions and create visually appealing web pages. With my expertise in coding, I can effectively tackle complex programming tasks and deliver high-quality results.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The Wilkinson catalyst chlorotris (triphenylphosphine) rhodium(I), ClRh(PPh0)3, brings about the catalytic hydrogenation of an alkene in homogeneous solution: (a) Using the following mechanistic...
-
For the complexes shown here, which would have the d electron distribution shown in the diagram below: [MF6]3, [M(CN)6]3, [MF6]4, [M(CN)6]4? Note that the neutral metal atom, M, in each complex is...
-
The acetylacetone ion forms very stable complexes with many metallic ions. It acts as a bidentate ligand, coordinating to the metal at two adjacent positions. Suppose that one of the CH3 groups of...
-
Mickey Limited is a manufacturing business that uses a standard costing system. The companys flexed budget for April 20X9 is: Sales 173,340 Costs Direct materials (22,500) Direct labour (37,684)...
-
Data for Hubble Company are presented in P12-3B. Instructions Prepare the operating activities section of the statement of cash flows using the direct method.
-
A section of duct system consists of 38 ft of straight 12-by-20-in rectangular duct, a wide-open damper, three smooth 90 elbows, and an outlet grille. Compute the pressure drop along this duct...
-
What is the plain-meaning rule?
-
Martin Buber Co. purchased land as a factory site for $400,000. The process of tearing down two old buildings on the site and constructing the factory required 6 months. The company paid $42,000 to...
-
what are three techniques for processing BI DATA? explain clearly REPORTING ANALYSIS, RFM ANALYSIS,ONLINE ANALYTICAL PROCESSING,DATA MINING ANALYSIS?
-
Comment on the following observations: (a) In the IR spectrum of free MeCH=CH 2 , C=C comes at 1652 cm 1 , but in the complex the corresponding absorption is at 1504 cm 1 . (b) At 303 K, the 1 H NMR...
-
(a) The reaction between BiCl 3 and 3 equivalents of EtMgCl yields compound X as the organo product. Two equivalents of BiI 3 react with 1 equivalent of X to produce 3 equivalents of compound Y. In...
-
Did various groups use the same technique in different ways or to accomplish different goals?
-
4. Air with initial conditions of 0.14 m, 66 C, and 500 kPa is expanded and ends up at 2 C. No heat is transferred during the expansion. What is the mass and what is the final volume? (0.719 kg,...
-
An airplane weighing 5,000 lb is flying at standard sea level with a velocity of 350 mi/h. At this velocity, the L/D ratio is a maximum. The wing area and aspect ratio are 200 ft2 and 8.5,...
-
Q4.) The circular cross-section of a pipe is given by diameter D = 10 cm, where the friction factor f = 0.065. If the flow is entirely turbulent, what is the surface roughness of the pipe? Be sure to...
-
1. Consider an aircraft designed for a positive limit load factor of 3.5. a) What is the structurally safe minimum radius of a symmetric pull out that the aircraft can execute at a dive speed of 350...
-
Air at 30C flows over a flat plate 1.5 m long and 1 m wide. The plate surface is maintained at 70C. If the desired rate of heat dissipation from the plate is 3 kW, determine the flow velocity of the...
-
Lark Corporation is considering the acquisition of an asset for use in its business over the next five years. However, Lark must decide whether it would be better served by leasing the asset or...
-
The financial statements of Eastern Platinum Limited (Eastplats) are presented in Appendix A at the end of this textbook. Instructions (a) Does East plats report any investments on its statement of...
-
a) NaOH is a strong nucleophile and strong base. The substrate in this case is primary. Therefore, we expect S N 2 (giving the major product) and E2 (giving the minor product). b) NaSH is a strong...
-
Calculate w, q, H, and U for the process in which 1.75 mol of water undergoes the transition H 2 O(l, 373 K) H 2 O(g, 610.K) at 1 bar of pressure. The volume of liquid water at 373 K is 1.89 10 5 m...
-
Identify the mechanism(s) expected to operate when 2-bromopentane is treated with each of the following reagents: a) NaOEt b) NaI/DMSO c) DBU d) NaOH e) T-BuOK
-
A solution contains an abundance (i.e., a lot) of organic carbon (in the form of acetate, CH 3 COO- ), with COD that far exceeds the concentration of all electron acceptors in solution. In addition...
-
Farmer Michael is developing a process in a batch reactor which can produce a new fertilizer compound. By taking secret ingredient, A, and running the reaction in a batch reactor, the decomposition...
-
List the five ways companies can departmentalize. Then, give an example of each of the five.

Study smarter with the SolutionInn App


