Suggest products of the following reactions: Et0 (a) MeBr + 2Li (b) Na + (C6H5) 2 (c)
Question:
Suggest products of the following reactions:
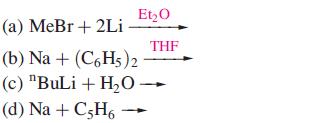
Transcribed Image Text:
Et₂0 (a) MeBr + 2Li (b) Na + (C6H5) 2 (c) "BuLi + H₂O → (d) Na + C5H6 THF
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a MeBr 2Li in Et2O ether This is a simple reaction of methyl bromide MeBr with lithium Li in diet...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the products of the following reactions (assume that excess reducing agent is used in d): a. b. c. d. CCH3 1. NaBHa 2. H20 COCH3 H2 Pt CH2COCH3 1. NaBHA 2. H20 CH2COCH3 1LiAlH4 2. H2O
-
Give the products of the following reactions a. b. 1. CH3O 2. HC CH CH CH COCH 1. CHaCH20 2. HCI CH,CHCH COCH CH3 CH3
-
Give the products of the following reactions and their configurations: HBr peroxide Br2, CH2CI2 CH3 0 Br2, H2o
-
Harvold Company's quality cost report is to be based on the following data: Test and inspection of incoming materials. $71,000 Supplies used in testing and inspection . Re-entering data because of...
-
The following T-account is a summary of the cash account of Kemper Company. What amount of net cash provided (used) by financing activities should be reported in the statement of cashflows? Cash...
-
Light reflected from a glass (n g = 1.65) plate immersed in ethyl alcohol (n e = 1.36) is found to be completely linearly polarized. At what angle will the partially polarized beam be transmitted...
-
Cat Auto Tech. Corp. purchased 10,000 gift certificates from DeJesus. Cat Auto Tech. Corp., an Amoco gasoline station operator, contracted with DeJesus to make 10,000 gift certificates of various...
-
The Shop at Home Network sells various household goods during live television broadcasts. The company owns several warehouses to hold many of the goods it sells, but also leases extra warehouse space...
-
1. A thin film is laid over a glass pane as shown. White light is incident on the film, coming straight in. At a point where the light is incident on the film, it appears green ( = 525 nm). Find (a)...
-
Measures taken to control atmospheric pollution include (a) Scrubbing industrial waste gases to remove SO 2 , (b) Reduction of NO in motor vehicle emissions. Explain how these are achieved and write...
-
Summarize the operation of a three-way catalytic converter, including comments on (a) The addition of cerium oxides, (b) The light-off temperature, (c) Optimum airfuel ratios (d) Catalyst ageing.
-
What is meant by CVP analysis?
-
Boheme's accountant is advised that Rhapsody, a trade receivable (debtor) for 2,300 is bankrupt and no payment is likely. Rhapsody is included in Receivables above. It is usual for businesses such as...
-
Southwest Co. borrows $100,000 on January 1 from Northeast Bank at a 12% interest rate. Southwest assigned $140,000 of its accounts receivable as collateral, and agreed to pay a financing fee of 2%...
-
What does architecture represent in the poem beowulf ?
-
Problem #9 The 12 annual deposits shown were made into an account paying 6% annual interest. 34 5 3. 6 34 34 fe * N L 12 13 H 20 mm.com The equivalent worth of the deposits in year 2 is closest to:
-
Problem 4 Roland had revenues of $600,000 in March. Fixed costs in March were $200,000 and profit was $40,000. Answer the following questions: a. What was the contribution margin percentage? b. What...
-
Julie is considering three alternative investments of $10,000. Julie is in the 28% marginal tax bracket for ordinary income and 15% for qualifying capital gains in all tax...
-
Gordon and Lisa estimate that they will need $1,875,000 in 40 years for their retirement years. If they can earn 8 percent annually on their funds, how much do they need to save annually?
-
The products obtained from hydroboration-oxidation of cis-2-butene are identical to the products obtained from hydroboration-oxidation of trans-2-butene. Draw the products and explain why the...
-
As the pressure is increased at 45C, ice I is converted to ice II. Which of these phases has the lower density?
-
Compound A has molecular formula C 5 H 10 . Hydroboration-oxidation of compound A produces an alcohol with no chirality centers. Draw two possible structures for compound A.
-
Calculate the following derivative if h(w) = wew (Use symbolic notation and fractions where needed.) h(2)(1) =
-
Consider an object moving along a line with the given velocity v Assume t is time measured in seconds and velocities have units of a. Determine when the motion is in the positive direction and when...
-
What types of internal corporate communication involves managing internal memos, emails, and monthly and annual reports while providing surveys and responding to internal audits? Discuss each type in...

Study smarter with the SolutionInn App


