The 129 Xe- and 19 F-NMR spectra of [XeF 3 ] + are shown in Fig. 8.56.
Question:
The 129Xe- and 19F-NMR spectra of [XeF3]+ are shown in Fig. 8.56. Interpret these spectra in light of the shape of [XeF3]+ predicted from VSEPR theory.
Figure 8.56.
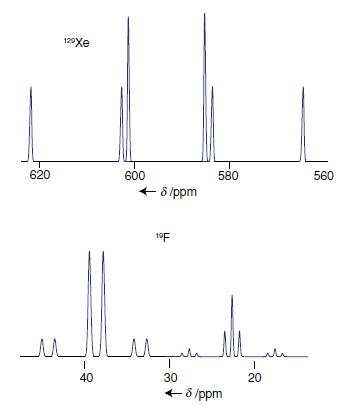
Transcribed Image Text:
620 سد 129Xe | 40 600 ← - 6 /ppm 10F 580 سل السيد بلد ا 30 ← 8/ppm 20 560
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The NMR spectra of 129Xe and 19F for XeF3 provide valuable information about the molecular structure of XeF3 and the chemical environment of the xenon ...View the full answer

Answered By

User l_998468
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The 1H NMR spectra of three isomers with molecular formula C7H14O are shown here. Which isomer produces which spectrum? a. b. c. QUESTION CONTINUE TO NEXT PAGE 0 (ppm) frequency 6 (ppm) frequency 8...
-
The 1H NMR spectra of two carboxylic acids with molecular formula C3H5O2Cl are shown in Figure 14.14. Identify the carboxylic acids. (The "offset" notation means that the signal has been moved to the...
-
In our development of consumer theory, we made a big point about the fact that neoclassical economics does not put much stock in the idea of cardinally measuring utility (in terms of units of...
-
A block of ice with mass 2.00 kg slides 0.750 m down an inclined plane that slopes downward at an angle of 36.9 below the horizontal. If the block of ice starts from rest, what is its final speed?...
-
Bailey Cleaners has the following balance sheet items. Accounts payable ........Accounts receivable Cash ...........Notes payable Equipment ..........Salaries and wages payable Supplies...
-
Researchers surveyed 19,183 U.S. physicians, asking for the information below. Location (region of the U.S.) income (dollars) employment status (private practice or an employee) benefits received...
-
John and Jennifer Margeson entered into a contract to sell a weight-loss franchise business called Inches-A-Weigh to Theresa Artis. The parties memorialized their agreement in an Asset Purchase...
-
Answer each of the questions in the following unrelated situations. (a) The current ratio of a company is 5:1 and its acid-test ratio is 1:1. If the inventories and prepaid items amount to $500,000,...
-
Jacinta had a bad experience with an insurance agent when her house flooded. The insurance agent was generally unhelpful and rude. She found herself becoming distrustful of all insurance agents,...
-
Calculate the expected wavenumber for an ND stretch given that NH stretches are normally observed at 3400 cm 1 .
-
Discuss how you would carry out the following analyses and determinations: (a) Calcium levels in breakfast cereal, (b) Mercury in shellfish, (c) The geometry of BrF 5 , (d) The number and type of...
-
Because of the growth of high-powered, low-cost computing, wireless communications and technologies such as videoconferencing, many managers soon may not need to come into the office to do their...
-
The construction team has equipped a new spray machine to paintwalls. As is common with most spray guns, unsightly blemishes oftenappear because of improper mixtures or other problems. A...
-
Use do..while loop to redisplay the menu Display the following menu on the screen: MENU 1. Process 1 2. Process 2 3. Process 3 0. Exit Type a number 1, 2, 3 to select the Process: Read the number....
-
Empirical evidence on firms' capital structure choices shows that (1) firms operating in different industries have different debt ratios (leverage), and (2) within an industry firm leverage and...
-
Canadas national statistics agency, Statistics Canada, saysthat the land area of Canada is 9,094,000 square kilometers. Of this land, 4,176,000 square kilometers are forested. Choose asquare...
-
Suppose data set 4.5, 40.5, 10.5, ...., has mean 16.5 and variance 144. The data is measured in inches The data is then transformed to measure in feet instead of inches. One foot equals 12 inches....
-
Assume that eight electrons are placed into the allowed energy levels of a cubic box where two electrons can occupy each allowed energy level. (See Exercise 148 for the appropriate energy equation.)...
-
Suppose that you are part of a virtual team and must persuade other team members on an important matter (such as switching suppliers or altering the project deadline). Assuming that you cannot visit...
-
For which member of the following pairs of complexes would oct be the larger and why: (a) [Cr(OH 2 ) 6 ] 2+ and [Cr(OH 2 ) 6 ] 3+ ; (b) [CrF 6 ] 3 and [Cr(NH 3 ) 6 ] 3+ ; (c) [Fe(CN) 6 ] 4 and...
-
The electronic absorption spectrum of [Co(OH 2 ) 6 ] 2+ exhibits bands at 8100, 16000 and 19400 cm 1 . (a) Assign these bands to electronic transitions. (b) The value of oct for [Co(OH 2 ) 6 ] 2+...
-
Arrange the following ligands in order of increasing field strength: Br , F , [CN] , NH 3 , [OH] , H 2 O.
-
Research Assignment - LGL217 Due: 2/26/24 Midnight via Email To Instructor The goal of this Research Assignment is to allow each of you to become more proficient in using electronic research...
-
Consider the following abbreviated financial statements for Parrothead Enterprises: PARROTHEAD ENTERPRISES 2017 and 2018 Partial Balance Sheets Assets Liabilities and Owners' Equity Current assets...
-
Use the method of averages to find the approximate yield rate for the bond shown in the table below. The bond is to be redeemed at par. Face Value $25,000 Bond Rate Payable Semi-annually 10% Time...

Study smarter with the SolutionInn App


